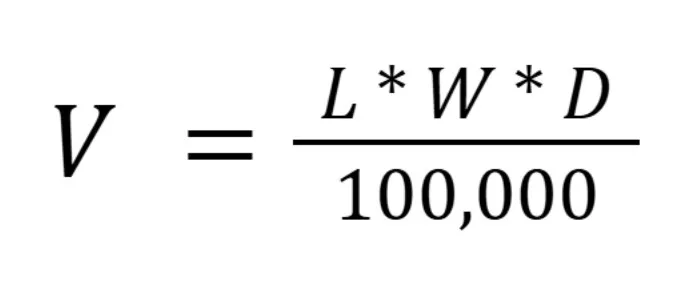Red tree vole nest site characteristics and persistence in young and old Douglas-fir forests: implications for canopy-dependent species
2 Summit Lake Paiute Tribe, Sparks, NV
3 National Council for Air and Stream Improvement, Inc., Corvallis, OR
JB: https://orcid.org/0000-0002-5646-5611
KM: https://orcid.org/0000-0002-3348-3403

Abstract photo. We conducted a red tree vole (Arborimus longicaudus) nest persistence study in Oregon’s Coast Range, 2019 – 2022. We conducted ground-based surveys in young forest stands (20 – 59 years) and canopy-based climbing surveys in older stands (60+ years) across 63 Douglas-fir dominated stands. Our key findings include nest persistence was primarily driven by nest size rather than forest age, with nests > 0.3 m³ achieving nearly 100% annual survival. Interspecific nest use between red tree voles and other canopy species was common in young forests, suggesting potential competition for limited nesting structures in young stands commonly managed for timber resources.
Abstract
Maintaining nest trees and enhancing structures in stands can support occupancy by canopy-dependent mammals, including at-risk species. Red tree voles (Arborimus longicaudus) are arboreal rodents that nest within the live crowns of trees, primarily in Douglas-fir (Pseudotsuga menziesii) forests of western Oregon and northwestern California. While abundance and distribution of arboreal nests in stands may inform likelihood of red tree vole occurrence or predict habitat, little is known about persistence or survival probability of natural red tree vole nests, especially in forests under 80 years of age. Further, understanding the relationship between nesting dynamics of canopy-dependent species, such as the red tree vole, and forest age characteristics may highlight pathways to improving wildlife habitat in timber-producing stands. To better understand red tree vole nest site characteristics, persistence, nest density, and interspecific nest use relationships, we randomly selected 63 Douglas-fir dominated stands in Oregon’s central Coast Range, stratified by stand age class (10-year increments from 20 – 50 years old, 60 – 79 years old, and > 80 years old) and distance (up to 5 km) from the nearest patch (> 20 ha) of forest over 80 years old, and latitude. We surveyed 6,557 trees across 63 stands for arboreal nests using plot-based ground and climbing surveys during 2019 – 2022. We recorded nest characteristics, status over time (present/absent) and evidence of arboreal species, including tree vole, use at 1,044 nests. We modeled nest persistence as a function of time, stand age, nest volume, nest construction type, and latitude using a known-fate modeling framework. Red tree vole nest persistence was highest (0.98, 95% CI = 0.81 – 0.99) in old forests (≥ 80 years old) where nests were more cryptic or associated with protective tree structures (e.g., cavities). Persistence was lower (0.9, 95% CI = 0.79 – 0.95) and more variable in young forest stands (<80 years old), and positively correlated with nest volume. Recently occupied tree vole nest density exhibited a bimodal pattern, with local peaks in 30-year stands (1.24 ± 0.35 nests/ha) within 1,425 m of old forest patches, declining to near-zero in 50 – 79 year stands, then reaching maximum densities in old forests (53.5 ± 14.9 nests/ha). In young forest stands, nests were often constructed and used by multiple species, most commonly red tree voles and Humboldt’s flying squirrel (Glaucomys oregonensis). In young forests stands, nests were often built on large, stable structures (e.g., split trunks and broken tops) compared to nests found in old forest stands, which were commonly supported by smaller structures (e.g. moss mats and epicormic branches) and used primarily by red tree voles. We described trends in nest persistence and interspecific nest construction, highlighting variations among nest availability, habitation, and interspecific interactions of red tree voles across forest age. Forest management practices in Oregon’s Coast Range that preserve and maintain diverse tree structures can enhance arboreal nest availability and longevity, supporting canopy-dependent species like tree voles. Our study may be especially relevant for habitat management in young forest stands where nesting space may be a limiting factor for red tree vole occurrence and reproduction.
Keywords: Arborimus longicaudus, tree vole, Douglas-fir, forest management, nest, Oregon, Pseudotsuga menziesii, survival
Introduction
For arboreal wildlife, the live crown, or the portion of a tree supporting living branches and foliage contributing to the forest canopy (Parker et al., 1995, Moffett, 2000), provides food resources, shelter from predators and the elements, space to reproduce and rear young and may facilitate dispersal (Carey, 1996, Berthier et al., 2012, Wilson and Forsman, 2013). Arboreal nest occurrence and retention can increase diversity, survival and immigration of canopy-dependent species (Michel and Winter, 2009, Berthier et al., 2012, Deeming et al., 2023). Thus, the establishment and persistence of nests and natural nest-supporting tree structures can increase the resiliency of canopy-dependent species and contribute to forest food- and nest-web diversity (Jones et al., 2009, Michel and Winter, 2009, Ibarra et al., 2020). For instance, bird and mammal densities in mature, mixed-conifer forests in British Columbia, Canada were positively associated with increased nest-site availability (Aitken and Martin, 2012). However, physical characteristics, interannual use and maintenance, and species interactions of arboreal nests by canopy-dependent mammals are largely unknown.
In western Oregon and northwestern California, USA, the full extent of their range, red tree voles (Arborimus longicaudus; hereafter, tree vole) are considered the most canopy-dependent mammal in Douglas-fir (Pseudotsuga menziesii) dominated forests (Carey, 1991). Tree voles are also classified as a ‘species of greatest conservation need’ in Oregon primarily due to their limited mobility and dispersal combined with habitat loss from wildfire and intensive forestry practices (Oregon Department of Fish and Wildlife, 2016). Tree voles feed almost exclusively on fresh Douglas-fir needles, from which they also obtain water (Forsman and Price, 2011), and live twig cambium (Maser, 1965), collecting small branches, primarily at night, and feeding at their nest throughout the day (Forsman et al., 2009). Tree voles rely on canopy-based nests for reproduction, rearing young, and protection and concealment from predators (Maser, 1965, Forsman et al., 2009, Swingle and Forsman, 2009). So long as they have access to the live crown for food and movement, tree voles can nest in a variety of places including in tree cavities, dense branch structures and in existing nests constructed by other species (Lesmeister and Swingle, 2017).
Because tree voles spend the majority of their life inside their nests, only leaving for an average of < 30 minutes nightly (Forsman et al., 2009), nest stability and placement is essential for survival and reproduction. However, tree voles are not the only rodent species that depend on the forest canopy in western Oregon to fulfill key functions of their life history. Humboldt’s flying squirrel (Glaucomys oregonensis), woodrats (Neotoma spp.), Douglas squirrel (Tamiasciurus douglasii) and Townsend’s chipmunk (Tamias townsendii) also commonly use forest canopies for nesting, foraging, and reproduction such that these species all use nests within the live crowns of trees (Maser et al., 1981, Carey, 1991, 1996).
A variety of stand- and tree-specific characteristics influence nesting habits of tree voles in young forests. Because tree voles are canopy-dependent, they utilize physical connections in the live crown to travel between trees for food and escape from predators (Carey, 1991). Branch and tree connections that create pathways throughout the canopy, along with the availability of nest-supporting features and pre-existing arboreal nests, improve habitat suitability for tree voles and other arboreal species in young forest (Linnell et al., 2018). These nest-supporting features, often referred to as ‘tree structures,’ play an important role in facilitating forest biodiversity (Bruce et al. 1985). The longevity, quantity, and characteristics of arboreal nests likely differ among forest stands of different ages (Berthier et al., 2012, Jiménez-Franco et al., 2018). Old Douglas-fir forests, especially those with trees > 200 years, exhibit increased structural complexity compared to younger, managed forests (Ruggiero et al., 1991). Red tree voles have been described as an old forest obligate (Corn and Bury, 1986, Carey, 1991, Gomez and Anthony, 1998, Linnell and Lesmeister, 2019), possibly due to a preponderance of research and management in older stands, primarily on public lands (USDA and USDI, 1994). However, young forest stands less than 80 years old can also develop nest-supporting tree structures, providing nesting space for canopy-dependent mammals such as flying squirrels, woodrats, chipmunks, and tree voles (Maser et al., 1981, Carey, 1991, Michel and Winter, 2009, Linnell et al., 2018). Despite the presence of tree structures in young forests, the structural complexity of old forest greatly outweighs that of young forest (Michel and Winter, 2009), which often have reduced canopy complexity and densities of nest-supporting structures, as well as increased structural variability among stands (Michel and Winter, 2009, Griffey et al., 2020). Stands with insufficient nest-supporting structures tend to be characterized by lower species occupancy, survival and reproductive success of canopy-dependent species (Berthier et al., 2012, Jiménez-Franco et al., 2018). The abundance and density of such structures and their long-term effects on tree vole populations in young forests are unknown.
Observed patterns in tree vole occurrence and distribution may reflect a lack of nesting opportunities in young forests (Linnell et al., 2018, Linnell and Lesmeister, 2020). Installing artificial nest platforms in the the live crown of trees at densities of 2 per ha in young forest stands in Oregon’s central Coast Range resulted in increased occupancy of red tree voles as well as other canopy-dependent species like Douglas squirrels and Humboldt’s flying squirrel (Linnell et al., 2018). Limited nesting space or availability of suitable locations for nest placement may lead to an increase in interspecific competition between canopy-dependent mammals. Although this interaction is well documented in bird species (Kempenaers and Dhondt, 1991, Ingold, 1994, Charter et al., 2016), studies on nest space competition between arboreal mammals are scarce (Linnell and Lesmeister, 2020).
In addition to availability of nest supporting structures, persistence of nests between seasons and especially through winter, may be critical to arboreal mammal diversity and occurrence, especially rodents, which utilize their nests for a wide range of functional roles such as reproduction, protection from predators and thermal insulation (Deeming, 2023). While most avian nests are rebuilt annually (Hansell, 2000), this investment may be too energetically costly for species that rely on nests year-round for critical functions. Although tree voles often use multiple nests within a home-range, nest longevity is still critical for tree voles due to their low vagility and high dependence on arboreal nests (Carey, 1996, Swingle and Forsman, 2009). High variability in nest persistence, often modeled similar to animal survival, has been reported for ground-dwelling small mammals, with an annual probability of nest destruction often > 70% (Millar, 2007). Persistence of arboreal nests used by Sonoma tree voles (Arborimus pomo) declined over time with a median expected persistence time of 28.6 months, and although Sonoma tree vole nest persistence rates did not differ by stand age class, both nest persistence and recolonization varied over time (Thompson and Diller, 2002). If this pattern occurs more broadly, we predict increased arboreal mammal diversity and occupancy in areas with increased arboreal nest abundance and increased nest persistence.
Improved understanding of how stand age, nest supporting tree structures, and nest size influence nest longevity and use may help forest managers assess arboreal mammal populations within stands. Both nest site availability and nest persistence influence population occurrence and persistence (Jiménez-Franco et al., 2018), and nest presence can be used as a proxy for species occupancy (Pierluissi and King, 2008). However, this assumption depends strongly on a species’ life history dynamics and nest persistence and use patterns. If, for example, nest occurrence is correlated with number of tree voles in a stand, ground surveys for arboreal nests could be used to assess tree vole occupancy or abundance. Correlations among nests, tree vole density and predicted habitat suitability in the Pacific Northwest are not clearly understood (Forsman et al., 2019, Rosenberg, 2019). If nests have high turnover, with unused nests diminishing in size or frequently eliminated by weather or other stochastic events, then nest density is likely to correlate with use. However, if nests are relatively persistent even when unused, then nest presence may not directly reflect occupancy, site use, diversity, or abundance of canopy-dependent forest mammals, including tree voles. Correlation between nests and individual tree voles is further complicated by imperfect detection of both tree voles and their nests (Forsman et al., 2016, Marks-Fife, 2016).
Similarly, accurate density estimates are fundamental for conservation planning, yet few studies have provided detection-corrected nest density estimates for red tree voles across forest development stages. Maser (1965) estimated 2.47 tree voles/ha in old forest, while Thompson and Diller (2002) found Sonoma tree vole nest densities of 3.40 – 6.21 nests/ha in 30 – 60 year stands, though neither study accounted for imperfect detection. Marks-Fife (2016) provided the first detection-corrected estimates (1.91 ± 1.2 occupied nest trees/ha) but lacked sufficient sample sizes across age classes to identify trends. The relationship between nest density and individual tree vole density remains unclear, as individual voles may use 1 – 7 nests (Swingle and Forsman, 2009, McCoy, unpublished data). Describing density patterns across forest age could inform habitat capacity and provide context to our nest persistence results.
To better understand relationships between forest age and arboreal nesting dynamics, specifically regarding tree voles, our objectives were to 1) quantify arboreal nest site characteristics (e.g., supporting structures, branch and tree connections, nest volume, use) in Douglas-fir dominated stands that differ in age, 2) model persistence of non-species-specific arboreal nests and tree vole nests, 3) provide an index of detection-corrected nest density based on our data, and 4) describe patterns of nest use by tree voles and other canopy-dependent mammals. We predicted nest characteristics and supporting tree structures would differ among forest age as forest composition changed and that, as a result of variations in nest site characteristics across stand age, tree vole nest persistence would be lower in young forest stands (< 80 years old) than in older forest stands. We also predicted that interspecific nest use would be higher in younger forest stands based on assumed limited availability of nest supporting structures.
Methods and Materials
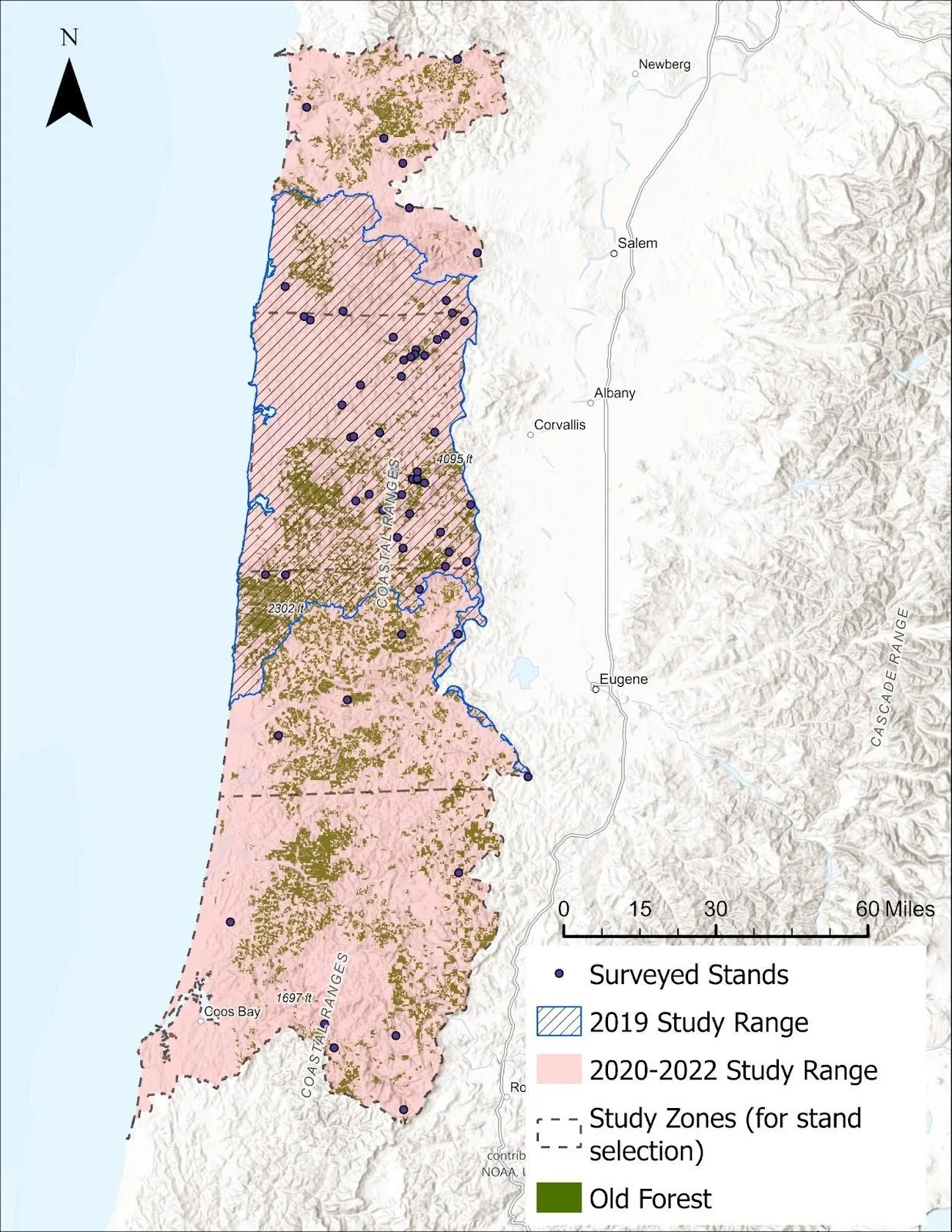
Our study evaluated arboreal nest site characteristics, persistence and density, with a specific focus on tree vole nests in the Oregon Coast Range, USA (study area – 12,466 km2, Figure 1). Throughout Oregon’s northernmost coast, where wildfire and timber harvest have constrained large areas of forest, tree voles are considered rare (Price et al., 2015, Linnell and Lesmeister, 2019, Linnell et al., 2023). Approximately 30% of our study area overlapped with the proposed Distinct Population Segment (DPS) of red tree voles, which had listing decisions under the U.S. Endangered Species Act (ESA) published in 2019 and 2023 (USFWS, 2019, 2023). The southern portion of our study area overlapped with areas of previous research aligning with more consistent sightings and occurrence (Forsman et al., 2016). Based on these previous surveys, we expected forest stands in the Oregon Coast Range to exhibit a gradient of tree vole occupancy probabilities and density, with the lowest occupancy rates assumed to occur at the northern extent of the species range, nearest to the Columbia River, likely due to intensive timber harvest and wildfire (e.g., Price et al., 2015, USFWS 2019, 2023, Linnell et al., 2023).
Before random sampling of stands was conducted, we created 3 stratifications to balance sample size among presumed tree vole density, stand age classes, and young forest stands with differing distances to old forest patches assumed to be inhabited by tree vole source populations. We defined a forest stand as a spatially contiguous community of trees that were similar in age, composition, and distribution such that they are distinct from adjacent tree communities (Nyland et al., 2016). We restricted sites to Douglas-fir dominated stands, but some multi-successional stands included western hemlock (Tsuga heterophylla) and Sitka spruce (Picea sitchensis) as co-dominant species, especially closer to the coast. In some unmanaged stands, grand fir (Abies grandis) and alder (Alnus sp.) were present. We restricted selected stand area to between 10 and 30 ha to balance sufficient stand area to harbor tree voles with the ability to conduct ground- and climbing-based activities in a reasonable timeframe. Additionally, we only considered stands if they were not scheduled to be harvested during the study period, and to mitigate bias resulting from habitat modification, we excluded stands that had been pre-commercially thinned. Our pilot season (2019) included a small extent of the central Coast Range, however in 2020, we evenly divided our study area into 4 latitudinal zones where we consistently stratified by stand age and distance to patches of old forest greater than 20 ha to ensure sufficient sampling across the full study area (Figure 1).
Young forest stands (< 80 years) in the Oregon Coast Range are commonly managed for timber production. We identified 7 landowners within our study and received inventory data specifying exact stand age. We randomly selected stands stratified by 6 age classes: 20 – 29, 30 – 39, 40 – 49, 50 – 59, 60 – 79, and > 80 years of age, aiming for at least 6 randomly selected stands of each class in each of the 4 latitudinal areas to evenly sample the entire study range.
To calculate the distance of young forest stands to the nearest patch of old forest, we created an old forest feature layer in ArcGIS (ESRI, 2020). We classified ‘old forest’ as stands > 80 years of age, aligning with established U.S. Forest Service definitions and previous tree vole studies (Old-Growth Definition Task Group, 1986, USDA and USDI, 1994, Marks-Fife, 2016, Rosenberg et al., 2016, Linnell et al., 2017). We downloaded and compiled LiDAR from the Oregon Department of Geology and Mineral Industries, collected between 2009 – 2012 (Downloaded 2015; https://www.oregon.gov/dogami/lidar/pages/index.aspx). We used stand age data from the Bureau of Land Management (BLM) Forest Operations Inventory Vegetation Publication (Downloaded 2020; https://gbp-blm-egis.hub.arcgis.com/datasets/6dd6576c4dd84197b3781938123e2d1b) to correlate LiDAR derived mean tree heights (m) for all inventoried stands > 80 years of age. Using this height-age association, we delineated our old forest layer to only include areas of trees > 42 m in height, subsequently assuming a relative stand age of > 80 years. We removed small patches of trees (e.g., riparian corridors), only including patches of old forest ≥ 20 ha in size, the minimum area assumed for self-sustaining tree vole populations based on density estimates in old forest (Maser, 1965, Marks-Fife, 2016, Linnell et al. 2017). We cross-referenced our map with the Forest Activity Electronic Reporting and Notification System maintained by the Oregon Department of Forestry (https://ferns.odf.oregon.gov/) to eliminate areas that had been disturbed or harvested since LiDAR was collected. We referenced our map to a previous delineation of old forest for tree voles that used similar methods and > 20 ha patches developed by Linnell et. al. (2017) and visually evaluated areas using publicly available satellite imagery, eliminating patches that had been disturbed. Lastly, during field work, we verified the age of older stands by collecting data at random plots (1/ha density) using visual aging techniques outlined in Van Pelt (2007) and averaged our estimated field-verified age within each stand.
Within each randomly selected stand, we generated circular 17.8 m radius (995m2) survey plots at a density of one per ha using the random point tool in ArcGIS. These plots provided a minimum survey area of 10% of the total stand area. In forest stands aged 20 – 59, we conducted ground-based surveys of each plot for arboreal nests. During these surveys, we walked the entire plot area and used binoculars to comprehensively search the canopy for arboreal nests. We also thoroughly searched the canopy for nests from the ground while traveling between plots to increase the sample size of nests surveyed year after year for analysis of nest persistence. We uniquely marked all nest trees found with numbered aluminum tree tags and flagging. We recorded tree and live crown metrics and geolocation using Trimble Smart Receivers (Trimble Inc. Model R-1000 #99133). Following our ground survey, we climbed all trees with nests discovered in the live crown that we were able to safely climb using spur climbing techniques (Berdeen et al., 2015).
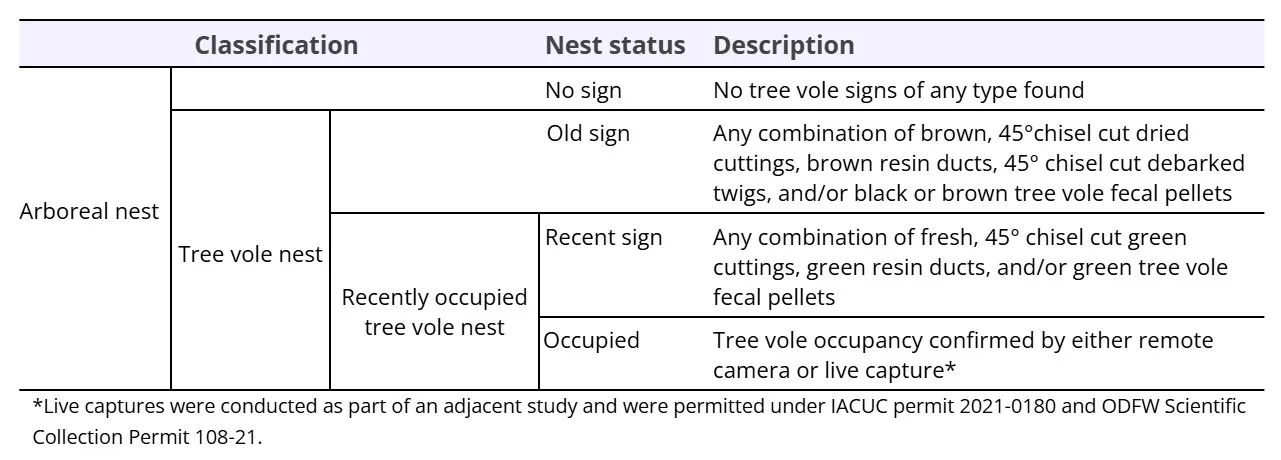
During initial site evaluation in stands over 60 years old, we determined that ground surveys likely would be ineffective given height and complexity of the canopy, aligning with prior research which reported nest tree detectability from the ground to be as low as 7% in mature (80 – 200 years) and 3% old (> 200 years) forests (Marks-Fife, 2016). Therefore, instead of conducting ground-based surveys in stands over 60 years of age, we searched plots for nests from within the canopy using a new vertical-climb survey method which we developed. Upon entering each randomly generated plot, we selected a tree within the plot to represent the ‘center.’ We selected this tree based on climbing safety, crown-to-crown visibility to other trees within the plot, and presence of tree structures that could potentially support tree vole nests. Once selected, we climbed the plot center tree using stationary rope climbing techniques (Berdeen et al., 2015). We surveyed for nests within the plot center tree and in the live crown of surrounding plot trees. Following this, any nests that we discovered in adjacent trees were then climbed and inspected for evidence of tree vole presence (Table 1).
Nest site characteristics
During nest surveys, we examined physical evidence (e.g., nest composition, materials) to determine nest construction and use by tree voles and other arboreal species. We determined tree vole nest status by searching each nest for presence of 45° chisel-cut Douglas-fir cuttings (between 1 – 35 cm long), discarded resin ducts stripped from fir needles during feeding, 45° chisel-cut debarked twigs (< 6 cm long), and tree vole fecal pellets (Lesmeister and Swingle, 2017). We classified tree vole nests as ‘old’, if resin ducts, fecal pellets, and collected Douglas-fir cuttings were dried and brown, or ‘recent’ if resin ducts, fecal pellets, and collected Douglas-fir cuttings were fresh and green (Table 1). We examined nests for evidence of other arboreal species. For example, fecal pellets and long Douglas-fir cuttings (> 30 cm long) associated with Bushy-tailed woodrat or fresh moss and bark shavings associated with Humboldt’s flying squirrel (Carey, 1991, Maser, 1998, Lesmeister and Swingle, 2017).
Accurately evaluating nest use was not possible via ground-based observation and required climbing to visually inspect nests within the live crown. To permanently identify nests, we marked each nest at nest height with a uniquely numbered aluminum tag painted brown to minimize the risk of attracting nest predators. We quantified the physical characteristics of each nest and the type of tree structure that physically supported each nest. We identified tree structures, which were often the result of abnormal growth patterns or a prior injury or infection to the bole or branches, using categories established in Swingle (2005), Michel and Winter (2009), and Marks-fife (2016) (Supplemental Table S1).
We estimated nest volume (V) in meters cubed using the volume calculation:
Where L was nest length in cm measured perpendicular to the bole of the tree, W was nest width in cm measured parallel to the bole of the tree, and D was nest depth in cm measured vertically from the top to the bottom of the nest.
To evaluate live crown connectivity at nest sites, we quantified the number of branch connections and tree connections for each nest. We counted physical branch connections to other live conifer trees within one branch whorl above and below the nest. We counted tree connections as the total number of live conifer trees with a direct physical connection to the nest tree.
To assess the availability of tree structures in each stand, we counted the total number of each structure type encountered in each plot. In young forest stands, we counted tree structures during ground surveys. In old forest stands, where it was impossible to visually survey an entire tree from the ground, we verified and quantified the occurrence of available tree structures through climbing. Since our plots represent 10% of the stand area, we extrapolated our tree structure counts to the stand level to create an estimate of tree structure density in each stand.
To identify temporal patterns in interspecific nest use, we estimated the number of distinguishable nest layers corresponding with evidence of how one or more species may have used the nest over time. We assigned a species to each nest ‘layer’ using evidence as being built or used by a particular species similar to previous research (Maser et al., 1981, Carey, 1991, Maser, 1998, Lesmeister and Swingle, 2017). For example, a nest may have initially been constructed and used by Humboldt’s flying squirrel, evidenced by the presence of a single moss-lined chamber about the size of a grapefruit (Maser et al., 1981). The same nest may have later been colonized by a tree vole, as evidenced by presence of discarded resin ducts and debarked twigs (Lesmeister and Swingle, 2017). Because arboreal species may colonize and add nest material on top of or inside existing nests, spatial placement of nest material may not coincide with chronological nest construction or use. Thus, we quantified and classified nest layers on a temporal scale such that the newest layer was attributed to the species that left the most recent sign of construction or habitation. For instance, if a nest exhibited recent tree vole sign, we attributed the most recently constructed layer to this species (Supplemental Figure S1). We associated suspected species to individual nest layers only when able to determine distinct signs of species-specific nest material or construction habits. We classified ‘undetermined’ nests as nests or nest layers with unidentifiable construction characteristics.
Estimating nest density
We provided a density estimation of both arboreal nests and recently occupied tree vole nests using our plot-based nest data and detection rates derived from double-sampling in young forests (p = 0.84, 95% CI 0.72 – 0.96) and census surveys in old forests (p = 0.055, 95% CI 0.00 – 0.12; details in Piasecki, 2023). Briefly, in old forest, we applied the Lincoln-Peterson abundance estimator to determine detection probability (p) for each plot: N = (n/p), where n represented nests found within plots during initial survey, and N represented all nests found in plots during census survey. We calculated density estimates by dividing relative abundance above by area in ha. We generated 95% confidence intervals by extrapolating confidence bounds of detection probabilities.
Estimating nest persistence
We revisited arboreal nests annually after their initial discovery and climbed trees with newly observed nests to document new construction and colonization. We evaluated nest persistence for (1) arboreal nests, which we classified as a nest structure in the live crown of the tree constructed or inhabited by any species (bird or mammal), and (2) tree vole nests, which we defined as any arboreal nest that exhibited any tree vole sign at any survey occasion from 2019 to 2022. We classified nests as ‘present’ (1) if they could provide suitable shelter, and nests that had been destroyed or decomposed to a state where they would be functionally ineffective as ‘absent’ (0) (Figure 2). If a nest was classified as ‘absent’, we did not resurvey the following year (i.e., we did not document rebuilding/recolonization at extirpated nest sites). Additionally, in a survival model framework, an individual cannot be classified as ‘present’ once it has been classified as ‘absent’. If a nest was unable to be surveyed in subsequent years or removed from the study due to climbing safety, it was assigned a censored value (.) for analysis.
We estimated nest persistence using a known-fate modeling framework in Program MARK (White and Garrott, 1990, Cooch and White, 2019). Under our definition of nest persistence as ‘present’ or ‘absent’, we treated all surveyed nests as marked individuals that were able to be monitored until they became extirpated (became absent), were censored, or reached the end of the study. Because we geolocated and marked nest trees and nests, the re-encounter probability of any nests found was 100%. We used binomial known-fate modeling to estimate annual probability of nest persistence, modeled as survival (S), for arboreal nests (Cooch and White, 2019). We modeled arboreal nest persistence across each annual time interval (i.e., year: t), stand age at the time of initial survey (stand age), and nest size in cubic meters at the time of initial survey (nest size). We modeled tree vole nest persistence in relation to t, stand age, and nest size. We also included nest construction type (construct) to determine if probability of tree vole nest persistence changed between nests originally constructed by other species and nests constructed by tree voles. Finally, we included latitude in decimal degrees (latitude) in our model for tree vole nest persistence. We predicted tree vole nest persistence would increase with higher presumed tree vole densities in the southern portion of our study area, assuming a positive relationship between tree vole density and frequency of nest construction and maintenance. We used Akaike’s Information criterion adjusted for small sample size (AICc), the difference between model AICc and the lowest model AICc in the model set (ΔAICc), and Akaike weights (AICc wts.) to determine the most parsimonious model (Burnham and Anderson, 2004). To evaluate the importance of covariates included in top-ranking models we considered those that were not zero (i.e., 95% CI did not overlap zero) as being strongly supported (Dugger et al., 2016).
Results
We surveyed a total of 63 stands from 2019 to 2022. Because of variation in funding, crew experience and staffing, and seasonal weather, our survey effort varied year-to-year. We surveyed 13 stands for 4 years, 20 stands for 3 years, 12 stands for 2 years, and 18 stands for 1 year (Supplemental Table S2). We surveyed 6,557 trees and found 1,044 arboreal nests located in the live crown that we investigated through tree climbing. We classified 564 (54%) as tree vole nests based on presence of old or recent tree vole signs (e.g., Table 1). The proportion of all arboreal nests with recent tree vole sign varied by year from 22% in 2019 (n = 12 stands with tree vole nests), 16% in 2020 (n = 28 stands with tree vole nests), 17.6% in 2021 (n=35 stands with tree voles nests), and 15% in 2022 (n = 40 stands with tree vole nests), our year with the most survey effort. We detected tree vole evidence within 61% of our stands (n = 39) varying between stand age class (e.g., 50% of our surveyed 20-year old stands within 1.4 km of old forest, 77% of > 80 year old stands), potentially suggesting that distributions were patchy across all age classes.
Nest site characteristics
We recorded nest dimensions and calculated nest volume in cubic meters (m3) for 1044 arboreal nests. The largest nest was 4.68m3 (200 cm W x 180 cm L x 130 cm D) and was initially constructed by a bald eagle (Haliaeetus leucocephalus). The smallest nest measured was 0.000192 m3 or 192 cm3 (8 cm W x 8 cm L x 3 cm D) and was initially constructed by a tree vole. Only 0.3% (n = 3) of all arboreal nests were larger than 1 m3, two of which were larger than 4 m3, highlighting that nests greater than 1 m3 were markedly rare. Mean nest volume across all age classes of forest stands was 0.042 ± 0.084 m3 while median nest volume was 0.016071 m3. Due to the presence of outliers in nest size, for comparative analysis, we focused on median nest volume, which was larger in young forest stand age classes relative to the old forest stand age class (Kruskal-Wallis rank sum test, p < 0.001) (Supplemental Table S3).
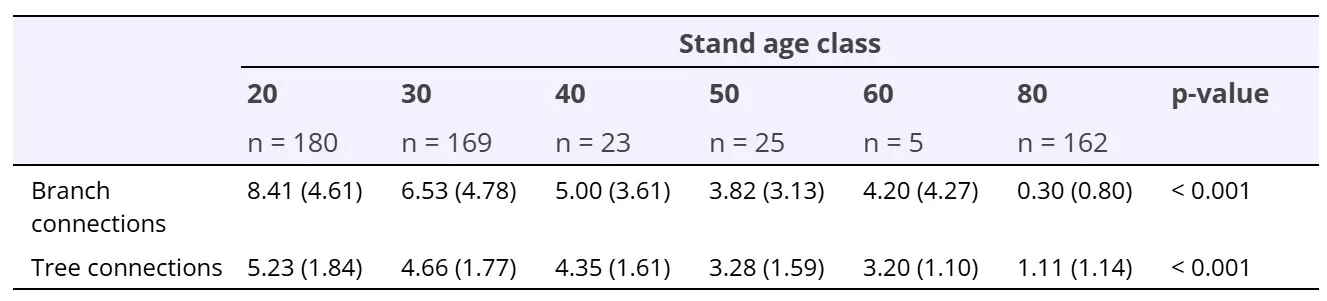
The average number of branch connections and tree connections at tree vole nests decreased as stand age increased (One-way ANOVA, p < 0.001; Table 2). We found 93.8% of tree vole nests in young forest had at least one branch connection within one whorl above and below the nest compared to only 16.7% of tree vole nests in old forest. We recorded 97.3% of tree vole nest trees in young forest with at least one tree connection to an adjacent live conifer compared to only 62.3% of tree vole nest trees in old forest.
Tree structure density increased with stand age and in almost all old forest stands, was an order of magnitude higher than in young forest stands (Figure 3). In young forest stands, most tree vole nest construction was supported by branch whorls, broken tops, and split trunks. For example, using data from 2022 (n = 564 nests), in the 20-year age class, these structures supported 46%, 28%, and 19% of tree vole nests respectively. In old forest stands, tree vole nest construction was supported largely by epicormic branch whorls (33%), large branches (25%), and moss mats (18%) (Figure 3; Supplemental Table S4)
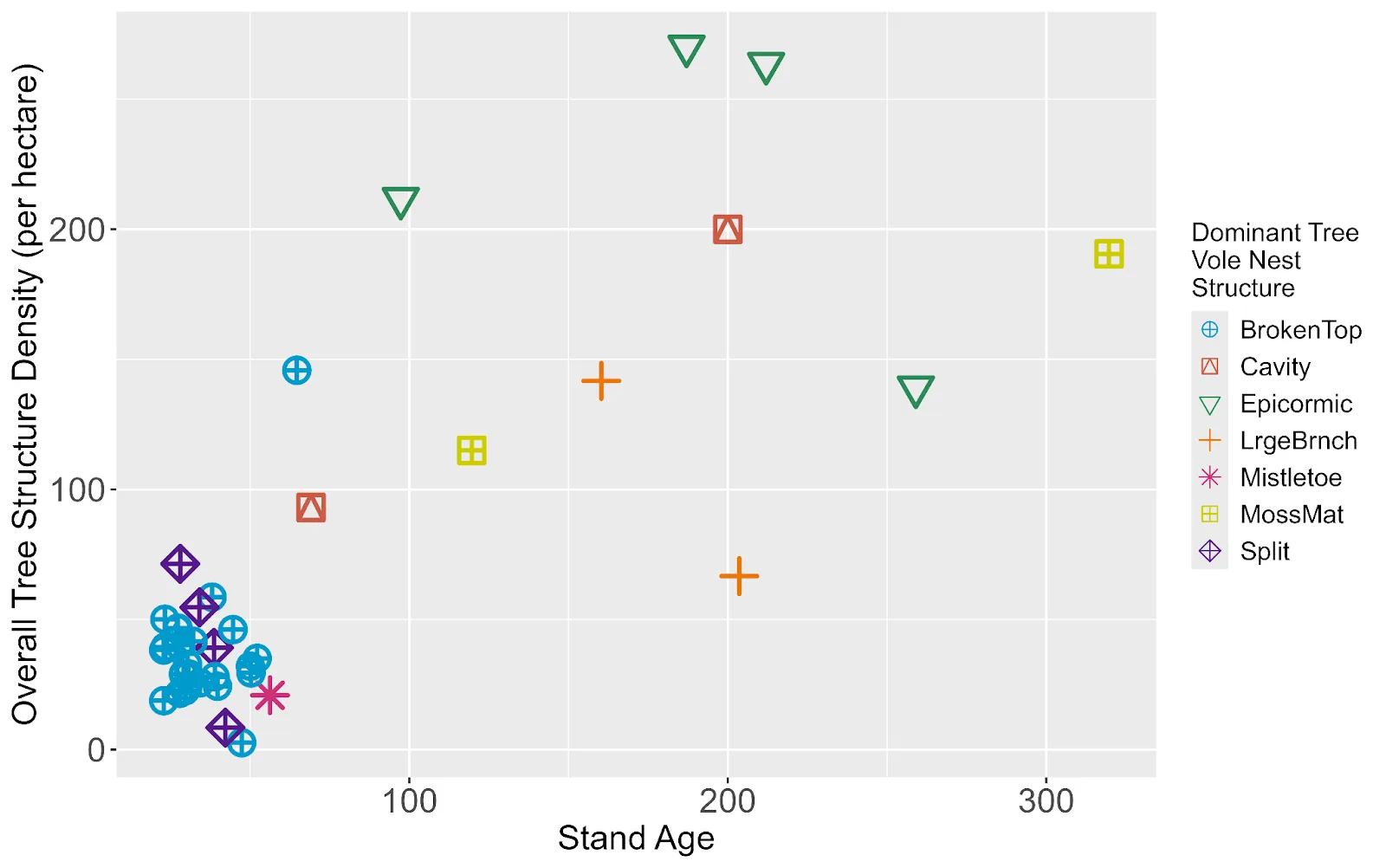
Figure 3. We observed varied tree structure density quantifying an estimate for all structure types (y-axis) across stand age. We represented the most common supporting tree structure for tree vole nests (shapes) in stands where tree vole nests were confirmed (n = 39). We excluded branch whorls as these have not previously been considered distinct ‘tree structures’ and their occurrence is ubiquitous across stand ages.
Tree structures that supported large arboreal nests varied with forest stand age. In old forest stands, where available, cavities (n = 19, 11%) supported the largest arboreal nests with a median volume of 0.06 m3 ± 0.17. In young forest stands, where cavities were not present, broken tops (n = 233, 27%) and split trunks (n = 154, 18%) supported the largest nests with a median volume of 0.02 m3 ± 0.31 and 0.02 m3 ± 0.04 respectively (Supplemental Figure 2).
Nest density
In young forests (20 – 59 years, n = 45 stands), arboreal nest density was highest in 20-year stands (3.76 ± 0.49 nests/ha) and declined linearly with age (R² = 0.31, p < 0.001). In old forests (≥ 80 years, n = 7 stands), density was an order of magnitude higher (152.9 ± 76.82 nests/ha) and increased with stand age, though confidence intervals were large due to low detection rates. In stands where recently occupied tree vole nests were detected, recently occupied tree vole nest density exhibited a bimodal pattern across age classes. Within young forests ≤ 1,425 m from old forest patches, which was the maximum distance from old forest that a tree vole nest was discovered, estimated recently occupied tree vole nest density peaked in 30-year stands (1.24 ± 0.35 nests/ha, n = 10 stands) and declined to near-zero in 50 – 79 year stands. In old forest stands, estimated recently occupied tree vole nest density was substantially higher (53.5 ± 14.9 nests/ha, n = 20) (Figure 4; Supplemental Table S5). Our maximum recorded nests per tree was 11 in old forests compared to 2 in young forests, indicating higher within-tree nesting capacity in structurally complex stand ages.
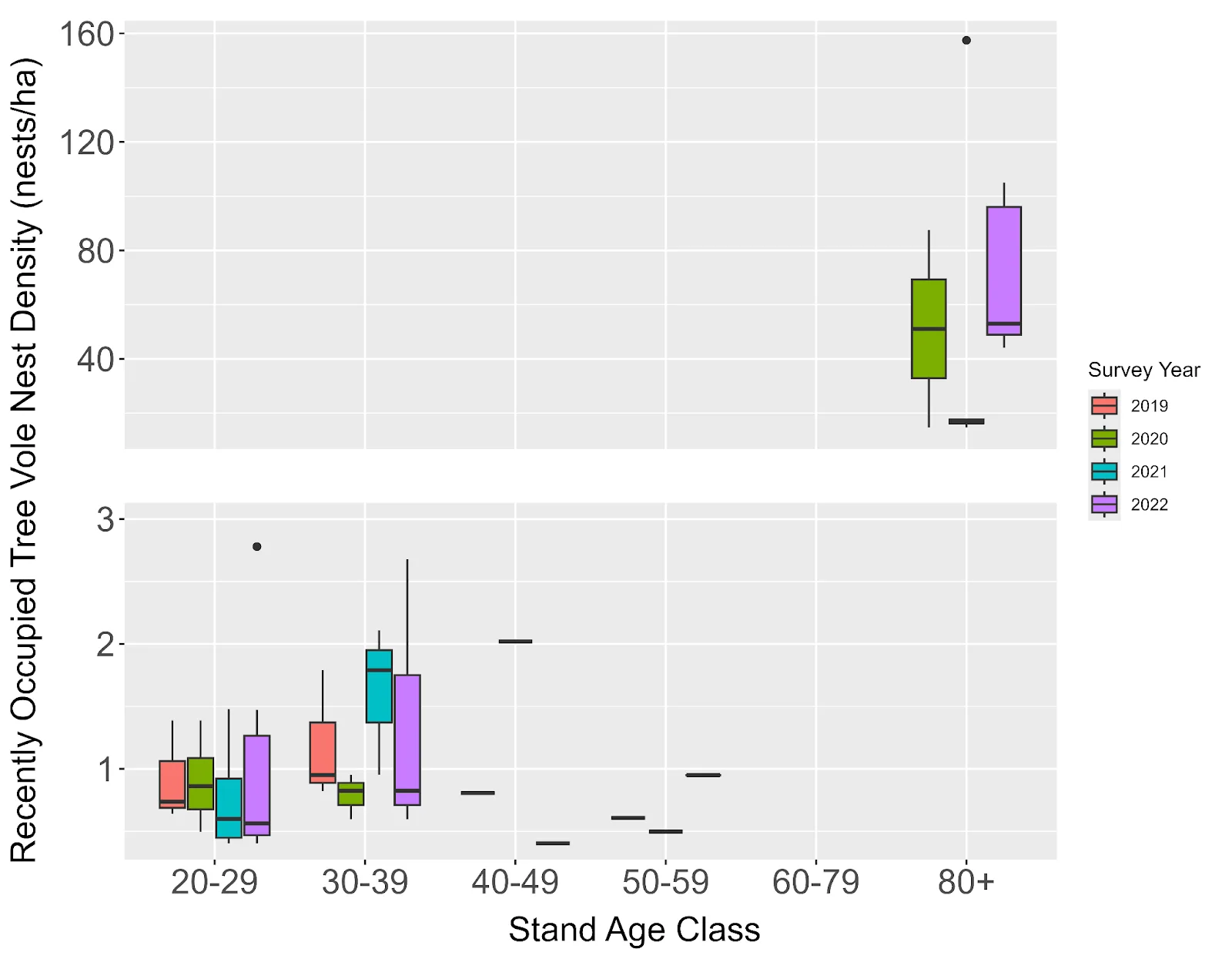
Figure 4. Estimated density of recently occupied tree vole nests for each age class in 2019 (red), 2020 (green), 2021 (teal), and 2022 (purple) using detection rates of 0.84 in young forests (0 - 80) years and 0.055 in old forest (≥ 80 years) forest (detection rates from Piasecki 2023). Excludes stands where no recently occupied tree vole nests were found. Sample sizes in Supplemental Table S5.
Arboreal nest persistence
We recorded nest persistence for 663 arboreal nests for at least 2 years between 2019 and 2022 (n = 980 observations). We observed nest extirpation across all age classes (Supplemental Table S6). The proportion of nests that became extinct was consistently highest in the 50- and 60-year age classes however, there were few nests found and surveyed for at least two years in these age classes (5% and 0.8% respectively).
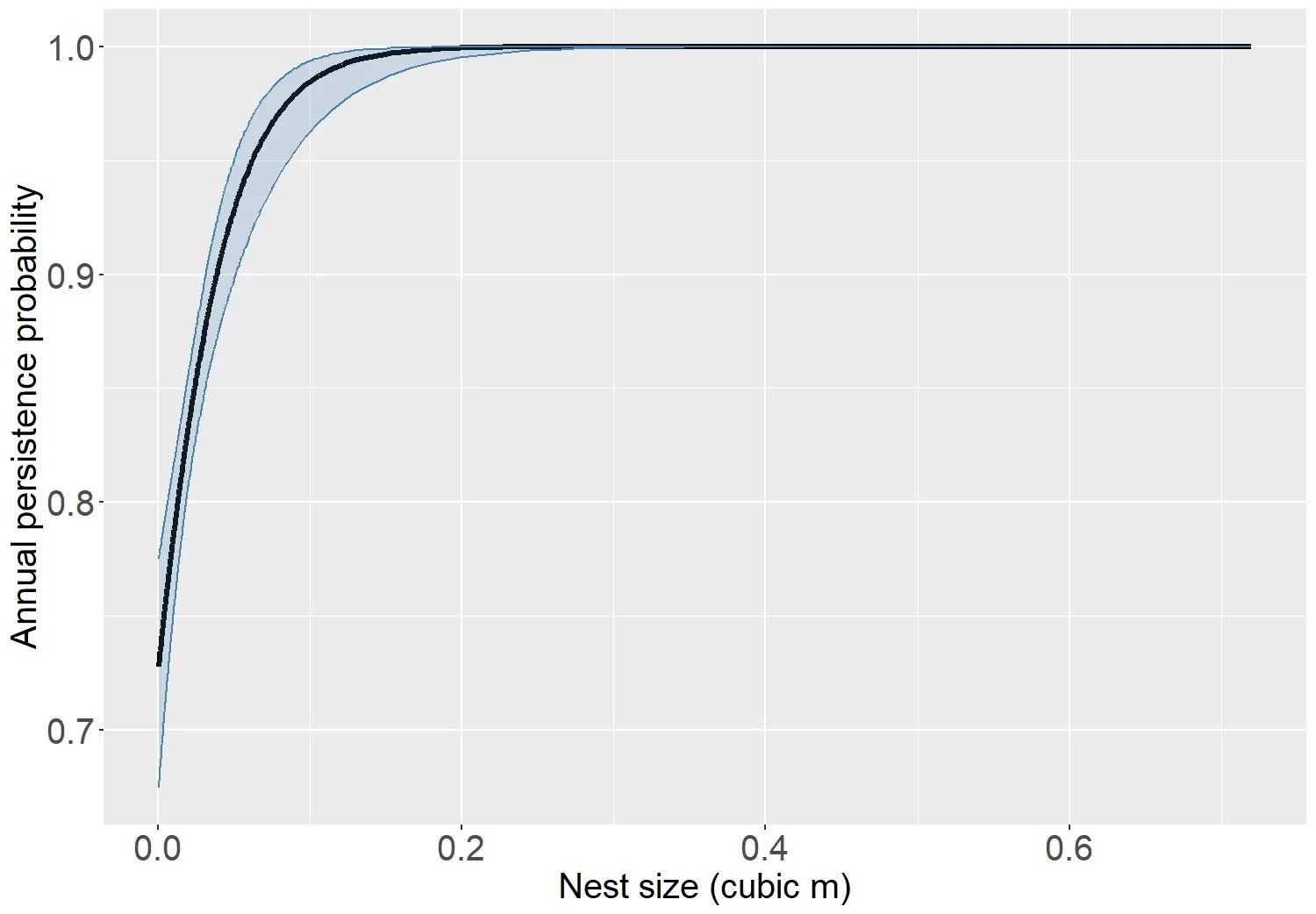
We modeled arboreal nest persistence using encounter histories over four annual survey occasions (2019 – 2022; 663 nests). The model with the strongest support included only nest size (S(nest size); AICc weight 61.78%, Table 3). Nest persistence increased linearly with nest size (βnest size = 31.8, 95% CI = 21.0 – 42.6), however when we graphed this relationship, it appeared as nest size increased, annual persistence probability approached an asymptote of near 100% (Figure 5). To more accurately capture this potential pseudo-threshold relationship, we added a post-hoc model that included the natural log of nest size (i.e., ln[nest size]). However, the model including ln(nest size) did not outperform the linear model suggesting the natural log of the nest size did not effectively account for enough variation in the data compared to the linear model (Table 3). This was likely because there were very few large nests > 1m3 , so while graphically these large nests appeared to have a strong influence on the survival threshold, in reality the remaining 99.7% of the nest size data was linearly associated with nest persistence.
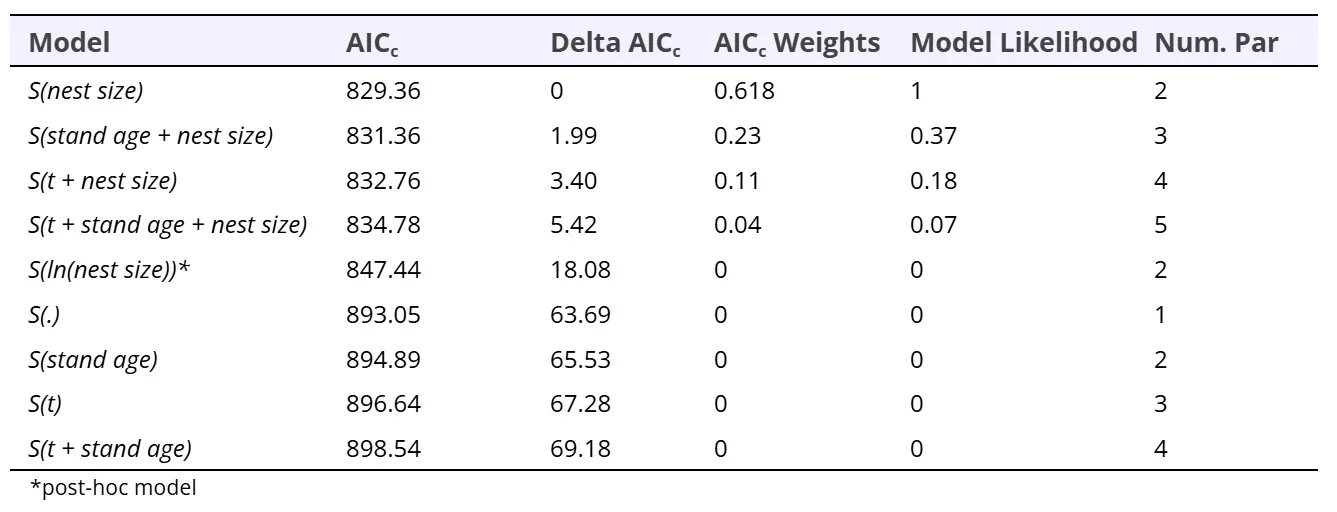
Table 3. A priori known-fate model selection results from analysis of annual persistence probability (S) of 663 arboreal nests across 3 time periods from 2019 – 2020, 2020 – 2021, and 2021 – 2022 (t) and/or nest size (continuous) and stand age (continuous). We listed models in order of increasing AICc values.
Tree vole nest persistence
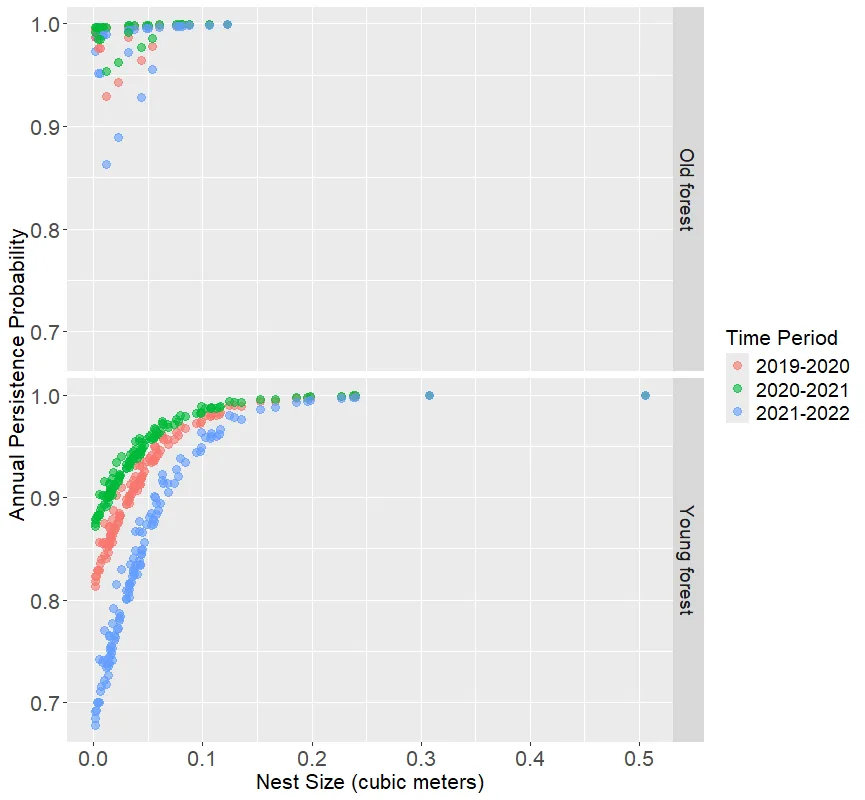
We also modeled nest persistence for tree vole nests with ‘recent’ or ‘occupied’ status that were surveyed for at least 2 years (2019 – 2022; n = 150 tree vole nests). Our top model included the additive effects of time interval, stand age, and nest size effects on annual persistence probability (Table 4). Nest size had a strong positive effect on annual persistence (βnest size = 22.3, 95% CI = 4.7 – 39.8), and stand age also had a positive but weak effect on annual persistence probability (βstand age = 0.01, 95% CI = 0.0 – 0.03) suggesting tree vole nests may be more likely to persist in older forests compared to young forests, however this determination would benefit from additional sampling. We observed some annual variation in nest persistence with higher persistence from 2020 to 2021 (βtime = 1.17, 95% CI = 0.03 – 2.32) relative to the reference year (2021 – 2022), but no difference in persistence from 2019 to 2020 given confidence interval limits that widely overlapped zero (βtime = 0.73, 95% CI = -0.44 – 1.90) (Figure 6, Table 4).
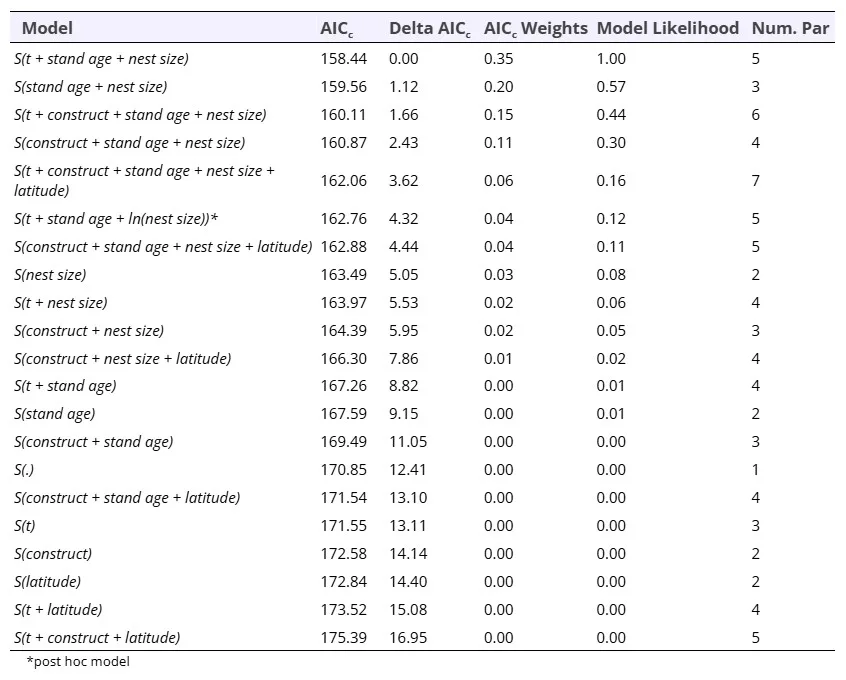
Table 4. A priori known-fate model selection results from analysis of annual persistence probability (S) of 150 red tree vole (Arborimus longicaudus) nests across 3 time periods (t) from 2019 – 2020, 2020 – 2021, and 2021 – 2022. We included an indicator for whether a vole or other species initially created the nest (construct; discrete), stand age (continuous), and latitude (continuous). Models are listed in order of increasing AICc values.
Our model suggested that, although weak, stand age influenced tree vole nest persistence such that annual persistence probability was lower (0.90, 95% CI = 0.79 – 0.95) and more variable (Range = 0.67 – 0.99) in young forests (<80 years) and higher (0.98, 95% CI = 0.81 – 0.99) and more stable (Range = 0.86 – 1.0) in old forests (> 80 years) (Figure 6). Using this model, the probability of a recently occupied tree vole nest of average size (0.04 m3) in a 35 year old forest persisting for 3 years was 0.74 (95% CI = 0.62 – 0.83), while the same sized nest in a 200 year old forest had a probability of (0.96, 95% CI = 0.66 – 0.99) to persist for 3 years. The relationship between nest size was similar for both tree vole and overall arboreal nest persistence where nests > 0.3m3 had very high likelihood (~1.0) of annual persistence (Figures 5, 6). Similar to modeling arboreal nest persistence we investigated the pseudo-threshold relationship between persistence and nest size by adding a post-hoc model including ln(nest size) (Table 4). Again, this model did not outperform the linear model because the logarithmic trend in the model only appears as a result of a single very large nest accounting for only 0.6% of the tree vole nest size data.
Interspecific nest use
We documented species associated with nest layer construction and/or use at every nest from 2020 to 2022. For summary, we highlight tree vole nests surveyed during 2022 (n = 439), which represents our largest annual sample size. Of nests with tree vole evidence, 72.2% (n = 317) were composed of a single distinct layer, 25.7% (n = 113) were of two identifiable layers, and 2% (n = 9) were of three identifiable layers. We identified multi-layer nests across all forest age classes but they were most common in 20- and 30-year age classes (44% and 33% of nests respectively) and least common in the > 80-year age class (4.3% of nests). Nests with 3 identifiable layers were only found in the 20- and 30-year age classes. Based on evaluation of nest layer data and suspected species associated with layer construction, tree voles and Humboldt’s flying squirrel exhibited the highest occurrence of interspecific nest use compared to other recorded species; 42.6% of all tree vole nests were originally constructed by Humboldt’s flying squirrel and 29.3% of Humboldt’s flying squirrel nests were originally constructed by tree voles (Table 5).

Table 5. Percentage of multi-layer red tree vole (Arborimus longicaudus) nests originally constructed by another arboreal species (n = 61) and multi-layer nests originally constructed by tree vole and later colonized by another arboreal species (n = 58) using data from 2022. We observed evidence of use by bushy-tailed woodrat (Neotoma cinerea), Humboldt’s flying squirrel (Glaucomys oregonensis), and tree squirrels (Tamiasciurus douglasii, Sciurus griseus). ‘Undetermined’ includes use but not characteristic evidence to a specific species or group.
We observed no temporally sympatric nest use between tree voles and other arboreal mammals. However, we documented green resin ducts, debarked twigs, and fresh Douglas-fir cuttings that were harvested by a tree vole pulled into a tree cavity directly under and extensively throughout the dense structure of a bald eagle nest. We observed a mating pair of bald eagles rearing chicks at this nest prior to climbing the tree to search for tree vole nests approximately two weeks after the chicks had fledged. Based on these observations, we suspected both species had been actively using the nest at the same time (Supplemental Figure S3).
Discussion
Through an extensive, 4-year study in Oregon’s central Coast Range, we found that arboreal nest persistence was largely dependent on nest volume and tree vole nest construction and use often overlapped with other canopy-dependent species in young forests. Through this large-scale effort, we developed an improved understanding of the drivers behind nest persistence and nest site characteristics for arboreal mammals in forests that differ in age.
Although nest persistence in young Douglas-fir stands was more variable than in old forest stands, nests had > 70% likelihood of annual persistence in most years especially for nests larger than 0.1 m3, which is similar to annual persistence estimates for Sonoma tree vole nests (Thompson and Diller, 2002, Figure 5), but significantly exceeds estimates for nests of ground-dwelling small mammals (Millar, 2007). Although prior research showed no significant difference of survival in nests in forest stands between 20 – 70 years (Thompson and Diller, 2002), we found some variability in nest persistence year-to-year in young forest stands. However, the main difference in annual nest survival was evident when comparing young forest stands to old forest stands. An average-sized nest in a 35-year-old stand had an estimated likelihood of 75% to survive for at least 3 years compared to 96% in a 200 year old stand. This is higher than the approximate 3-year persistence rate (45%) for Sonoma tree vole nests under average conditions in northwestern California, but this study did not account for nest size (Thompson and Diller, 2002). Our results suggest larger (> 0.3 m3) arboreal nests had higher annual survival rates with nearly 100% persisting in young forest stands from 2019 to 2022. Larger nests in our study were often supported by stable, robust tree structures, such as split trunks and broken tops in young forest stands and cavities in old forest. Our observations in young stands were similar to others where these tree structures trees with broken tops, split trunks or other defects in young stands bolstered construction of large nests, which were often used for reproduction and rearing of tree voles (Lesmeister and Swingle, 2017, Linnell et al., 2018). Thus, these structures may be essential stand-wide features for population persistence.
Although old forest stands exhibited increased diversity and availability of tree structures in the canopy compared to young forest stands, both stand age classes provided individual tree structures that supported arboreal nesting (Supplemental Table S4). In young forest stands, arboreal nests were often supported by platforms created by broken tops (e.g., presumed wind and snow breakage of the leading stem), split trunks (i.e., where two or more stems develop concurrently) and sites with dense, interconnecting branch whorls. Composition and location of broken tops and split trunks commonly resulted in high densities of interconnected live branches. Access to adjacent trees via these branch connections could facilitate movement throughout the canopy, and availability and persistence of these nest supporting structures could support occurrence and dispersal of tree voles and other canopy-dependent species in young forest stands.
In old forests, despite availability of split trunks and branch whorls, other tree structures characteristic of mature trees (e.g., cavities, moss mats, epicormic branches) were abundant and were used more often by tree voles to support nest construction. Cavities created by animal excavators (e.g., woodpeckers), fungi, or disease not only provided nest support but also likely increased concealment and protection from predators (Bunnell, 2013). Similarly, tree vole nests created under moss mats covering large branches, common throughout canopies of mature trees, were extremely difficult to locate and likely provided a high level of concealment from predators. Although we documented moss mat supported nests comprising 18% of tree vole nests in old forest stands (n = 162; 2022), these may be underrepresented due to their cryptic nature. Therefore, this nest type likely represents a larger proportion of nests than are represented by our data. Epicormic branch structures supported 33% (n = 53; 2022) of identified tree vole nests in older forest stands, consistent with previous findings (26%; Swingle, 2006).
Our observed bimodal pattern of recently occupied tree vole nest density across forest development stages provides some insights into availability. The local density peak in 30-year stands (1.24 nests/ha) likely reflects optimal conditions of high canopy connectivity (97.3% of nest sites had tree connections to adjacent tree crowns) combined with adequate structural complexity before stem exclusion begins to naturally thin the stand, reducing canopy connectivity and suitable nesting sites. The density decline in 50 – 79 year stands supports the hypothesis that this age class represents poor habitat due to reduced canopy connectivity and limited tree structure availability (Michel and Winter, 2009). This lack in nest density surrounding the 50 – 69 year age class was also observed in Sonoma tree voles (Thompson and Diller, 2002). The 43-fold difference in nest density between 30-year young forests and old forests (53.5 nests/ha) reflects fundamental differences in habitat quality and carrying capacity. Old forests supported up to 11 nests per tree compared to a maximum of 2 in young forests, indicating that tree structure availability enables higher within-tree occupancy. However, the relationship between nest density and individual tree vole density remains uncertain given that individuals can use multiple nests within a home range (Swingle and Forsman, 2009, McCoy, unpublished data).
As expected, arboreal nests in our study hosted a diversity of species. We documented interspecific nest construction and use by tree voles, Humboldt’s flying squirrels, bushy-tailed woodrats, other tree squirrels and a bald eagle (Supplemental Figure S3). Despite recording 4% of tree vole nests in young forest stands as having multiple distinct layers initially constructed by another arboreal species, sympatric nest use was rare and has only been documented at occupied tree vole nests with deer mice (Peromyscus maniculatus) and western grey squirrel (Sciurus griseus) (Brown, 1964, Maser, 1965). We suspect this behavior may occur in situations where nest size permits sufficient allocation of nesting space and where species interaction is limited and non-detrimental. We recorded the first occurrence of sympatric nest use by tree voles and bald eagles. Although bald eagles commonly prey on mammals, voles have not been extensively recorded in bald eagle diets (Jackman et al., 1999). These species may have co-habitated unnoticed due to tree voles’ small size and primarily nocturnal activity patterns. In this specific instance, the nest was located in a tree along the shoreline of the Alsea River which likely presented more abundant and optimal prey species than tree voles. Further, nests constructed by bald eagles offer protection and refuge from predators, structural stability, and because of their size, can fulfill space requirements for tree vole reproduction.
Interestingly, we recorded frequent interspecific nest use and construction between tree voles and Humboldt’s flying squirrel in young forest stands, while observing almost no overlap in nest construction or use between the two species in old forest stands. Multiple species using the same nests among years, or even months, may suggest limitation of desirable nest supporting tree structures, which likely results in competition for space. Nesting space has been predicted to be a limiting factor for occurrence and dispersal of tree voles in young forest stands (Linnell et al., 2018). Conversely, this observation may suggest a mutually beneficial relationship through interspecific nest construction and use where arboreal species benefit from pre-existing nests through a synergy for large-nest maintenance. For example, previous observations of female flying squirrels using ‘fragile’ nests for giving birth, especially in second-growth forests, was hypothesized to occur either because of competition over cavities or a preference towards ‘fresh’ structures (Carey et al., 1997). Flying squirrels have also been documented extirpating tree voles from occupied active nests and occasionally predating tree voles where nest use overlapped (Linnell and Lesmeister, 2020). Similarly, southern flying squirrels (Glaucomys volans) can be significant nest predators for birds (Degregorio et al., 2019). As such, we assume that flying squirrels may aid in nest persistence and maintenance of larger nests but would ultimately be detrimental to individual tree voles where nest use temporally overlaps.
In contrast to young forest, competition for nest space between tree vole and flying squirrel may be less prevalent in old forest stands as flying squirrels frequently nest in snags and cavities that are not available in younger stands in older stands (Carey et al., 1997). Snags and trees that lack forage opportunities (e.g., live branches) or connectivity to the surrounding forest canopy would likely be unattractive nest sites for tree voles. Further, overall availability of tree structures used for nesting was significantly higher in old forest stands than in young forest stands. While tree voles can construct their own nests on branch whorls and other structures, the energy requirements involved in nest construction and prolonged exposure to predators while nest building may be a significant trade-off (Guerra and Ades, 2002, Mainwaring and Hartley, 2013, Deeming, 2023). Therefore, in young forests, tree voles could be motivated to choose pre-existing natural nests (this study) or artificial nests (Linnell et al., 2018) if available, especially when those nests were large (> 0.3m3) and supported by stable tree structures.
While this study details the effects of stand age on nest persistence and nest site use, the scale at which we examined forest age as an independent variable varied between young forest stands and old forest stands. We balanced our study design, focusing on the range of forest ages 20 to 79 years (n = 54 stands). We combined old forest as all stands > 80 years, which spanned 97 to 320 years (n = 9 stands) and thus old forest surveys would benefit from additional replication across the range of ages included in our old forest age class to capture changes in tree crown and structure development throughout this category. Age estimation in old forest stands was challenging because older forest stands under natural succession were composed of trees from different age cohorts, so assigning a singular value to stand age did not capture the structural and developmental variability present. Finally, conducting effective surveys in old forest stands through our canopy-based survey method required significantly greater effort compared to surveys conducted in young forest stands and there is significant uncertainty in the overall distribution of voles in old forests. For instance, we observed vole sign in 7 of our 9 old stands (77%), which was significantly higher than Dunk and Hawley (2009) who reported 96 of 365 plots with vole sign, of which 91 plots with voles were in stands with tree diameters > 90cm (n ~258 late seral old growth plots; ~35% of the sample). Dunk and Hawley (2009) estimated the probability of Forest Inventory plot-level (1-ha, 100 x 100 m) red tree vole occurrence to be 25% in reserves and 17% for predicted habitat on public lands outside old growth reserves, given their data. Our inferences were strongest for 20- to 50-year-old stands, with less representation regarding the range of variation associated with arboreal and tree vole nest characteristics and persistence in older forest stands. Understanding vole occurrence would require additional effort following directed power analyses and modified sampling protocols (Piasecki, 2023, Moriarty et al., unpublished data). Further, we found that not every nest that exhibited recent tree vole sign was occupied. This is critical in informing future research and management regarding tree voles and aligns with results produced by Swingle and Forsman (2009) wherein a single tree vole can use multiple nests within its home range. Research that implements live-capture techniques to track tree vole movement and confirm individual nest use can effectively contextualize the relationship between recently occupied tree vole nests and abundance of individual tree voles within a stand.
Management Implications
Understanding persistence of arboreal nests can play an important role in conserving canopy-dwelling species and supporting their nesting requirements, especially in forests with limited structural complexity, where species may compete for nesting space. Our four-year study across Oregon’s central Coast Range demonstrated that arboreal nest persistence was primarily driven by nest size, with nests larger than 0.3 m³ achieving nearly 100% annual survival. While red tree vole nests in old forests (≥ 80 years) exhibited higher persistence rates than in young forests, the frequent interspecific nest use in young stands suggests young forests may provide habitat despite limited structural characteristics compared to old trees. Such young forest habitat is likely critical in facilitating connectivity for tree voles between patches of older forest refugia (Linnell et al., 2017). Given our high nest survival rates, caution is warranted when interpreting the relationship between nest abundance and red tree vole population size. In 2022, we documented recent tree vole sign in 43% of surveyed stands, with 15% of observed nests showing evidence of recent use and 20% of those classified as occupied. This translates to approximately 3 occupied nests per 100 nests in stands with confirmed tree vole occurrence, likely varying between young and old forest. We also highlight the necessity to consider differences in research methods in old forest compared to young forest accounting for challenges associated with canopy complexity and nest detectability. While these data provide initial insights into nest use variability, additional sampling and longer study duration are needed to clarify the relationship between nest density and individual tree vole abundance.
To support voles and canopy-dependent species, forest managers could prioritize canopy connectivity and retention of nest-supporting structures including broken tops, split trunks, and interconnected branch whorls during stand management operations to support construction of larger, more persistent nests and facilitate movement throughout the canopy. Given that 75% of average-sized nests in 35-year-old stands were likely to persist for at least three years, young managed forests can support small populations of red tree voles and facilitate connectivity between old forest patches when appropriate structural features are retained.
Because canopy research and monitoring, in particular assessments of nest use, is both physically challenging and requires specialized training and safety equipment, understanding the relationship among nest occurrence and persistence in relation to use, specifically by tree voles, would aid in assessing or prioritizing stands for surveys, management, and conservation. Future research and management efforts would benefit from further investigation into understanding tree vole demographic rates (i.e., reproduction, survival) in stands that differ in age.
Acknowledgments
The magnitude of this work was monumental and without the dedication and persistence of our field crews, none of this would have been possible. We graciously thank Mackenzie McCoy, Cody Berthiaume, Salix Scoresby, Jake Baker, Kaitlin Webb, Ian Shriner, Mark Stevens, Stephanie Loredo, and Jesse Ritter. We thank Eric Forsman, Jim Swingle and Mark Linnell for study design feedback and field assistance, and Katie Dugger, for invaluable contributions to modeling and manuscript review. We thank participating landowners and facilitators Jessica Homyack (Weyerhaeuser Company), Jenniffer Bakke (Manulife Investment Management), Fran Cafferata (Cafferata Consulting), Starker Forests, Hampton Lumber, Lone Rock Resources, Nick Palazzotto, Vanessa Petro and Corey Grinnell (Oregon Department of Forestry), Deanna Williams (US Forest Service), and Scott Hopkins (Bureau of Land Management). We also benefited from reviews by our participating landowner representatives, Darren Miller, Jake Verschuyl, our editor, and anonymous reviewers. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government. Protocols associated with determining vole nest occupancy were performed under protocols approved by Oregon State University’s Institutional Animal Care and Use Committee (IACUC) and the Oregon Department of Fish and Wildlife.
Author Contributions
Jason Piasecki: Conceptualization, data curation, formal analysis, methodology, project administration, writing – original draft
John Bailey: Conceptualization, supervision, writing – review and editing
Katie Moriarty: Conceptualization, funding acquisition, investigation, methodology, project administration, supervision, writing – review and editing
Data Availability
Field data were collected under research and/or confidentiality agreements and cannot be made publicly available. Please contact the lead author for data requests.
Supplementary Information
Supplementary information can be found here.
Transparent Peer Review
Results from the Transparent Peer Review can be found here.
Recommended Citation
Piasecki, J., J. Bailey, and K. Moriarty. 2026. Red tree vole nest site characteristics and persistence in young and old Douglas-fir forests: implications for canopy-dependent species. Stacks Journal: 26002. https://doi.org/10.60102/stacks-26002
References
Aitken, K. E. H., and K. Martin. 2012. Experimental test of nest-site limitation in mature mixed forests of central British Columbia, Canada: Nest-site limitation in cavity users. The Journal of Wildlife Management 76: 557–565. https://doi.org/10.1002/jwmg.286.
Berdeen, J., B. Chamberlain, T. Grubb, A. Henderson, B. Mayo, M. Mielke, K. Purcell, D. Ringnes, M. Roberts, D. Stubbs, and M. Thorning. 2015. National Tree Climbing Guide. Washington, DC: United States Department of Agriculture.
Berthier, K., F. Leippert, L. Fumagalli, and R. Arlettaz. 2012. Massive nest-box supplementation boosts fecundity, survival and even immigration without altering mating and reproductive behaviour in a rapidly recovered bird population. PLoS ONE 7: e36028. https://doi.org/10.1371/journal.pone.0036028.
Brown, L. N. 1964. Breeding records and notes on Phenacomys silvicola in Oregon. Journal of Mammalogy 45: 647. https://doi.org/10.2307/1377350.
Bunnell, F. L. 2013. Sustaining cavity-using species: Patterns of cavity use and implications to forest management. ISRN Forestry 2013: 1–33. https://doi.org/10.1155/2013/457698.
Burnham, K. P., and D. R. Anderson. 2004. Multimodel inference: Understanding AIC and BIC in model selection. Sociological Methods & Research 33: 261–304. https://doi.org/10.1177/0049124104268644.
Carey, A. B. 1991. The biology of arboreal rodents in Douglas-fir forests. Gen. Tech. Rep. PNW-GTR-276. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station. https://doi.org/10.2737/PNW-GTR-276.
Carey, A. B. 1996. Interactions of northwest forest canopies and arboreal mammals. Northwest Science 70: 72–78.
Carey, A. B., T. M. Wilson, C. C. Maguire, and B. L. Biswell. 1997. Dens of northern flying squirrels in the Pacific Northwest. The Journal of Wildlife Management 61: 684. https://doi.org/10.2307/3802176.
Charter, M., I. Izhaki, Y. Ben Mocha, and S. Kark. 2016. Nest-site competition between invasive and native cavity nesting birds and its implication for conservation. Journal of Environmental Management 181: 129–134. https://doi.org/10.1016/j.jenvman.2016.06.021.
Cooch, E. G., and G. C. White. 2019. Program MARK: A gentle introduction. 19th ed. Fort Collins, CO.
Corn, P. S., and R. B. Bury. 1986. Habitat use and terrestrial activity by red tree voles (Arborimus longicaudus) in Oregon. Journal of Mammalogy 67: 404–406. https://doi.org/10.2307/1380898.
Degregorio, B., J. Sperry, D. Kovar, and D. Steen. 2019. Southern flying squirrels (Glaucomys volans) as major predators of avian nest boxes in Conecuh National Forest, Alabama. Southeastern Naturalist 18: 476. https://doi.org/10.1656/058.018.0313.
Deeming, D. C. 2023. Nest construction in mammals: A review of the patterns of construction and functional roles. Philosophical Transactions of the Royal Society B: Biological Sciences 378: 20220138.https://doi.org/10.1098/rstb.2022.0138.
Dugger, K. M., E. D. Forsman, A. B. Franklin, R. J. Davis, G. C. White, C. J. Schwarz, K. P. Burnham, J. D. Nichols, J. E. Hines, C. B. Yackulic, P. F. Doherty Jr., L. Bailey, D. A. Clark, S. H. Ackers, L. S. Andrews, B. Augustine, B. L. Biswell, J. Blakesley, P. C. Carlson, M. J. Clement, L. V. Diller, E. M. Glenn, A. Green, S. A. Gremel, D. R. Herter, J. M. Higley, J. Hobson, R. B. Horn, K. P. Huyvaert, C. McCafferty, T. McDonald, K. McDonnell, G. S. Olson, J. A. Reid, J. Rockweit, V. Ruiz, J. Saenz, and S. G. Sovern. 2016. The effects of habitat, climate, and barred owls on long-term demography of northern spotted owls. The Condor: Ornithological Applications 118: 57–116. https://doi.org/10.1650/CONDOR-15-24.1.
Engeman, R. M., R. T. Sugihara, L. F. Pank, and W. E. Dusenberry. 1994. A comparison of plotless density estimators using Monte Carlo simulation. Ecology 75: 1769–1779. https://doi.org/10.2307/1939636.
ESRI. 2020. ArcGIS Desktop. Version 10.8. Redlands, CA: ESRI.
Forsman, E. D., and A. L. Price. 2011. Water consumption by red tree voles (Arborimus longicaudus). Northwestern Naturalist 92: 116–119. https://doi.org/10.1898/10-28.1.
Forsman, E. D., J. K. Swingle, R. J. Davis, B. L. Biswell, and L. S. Andrews. 2016. Tree voles: An evaluation of their distribution and habitat relationships based on recent and historical studies, habitat models, and vegetation change. PNW-GTR-948. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station. https://doi.org/10.2737/PNW-GTR-948.
Forsman, E. D., J. K. Swingle, and N. R. Hatch. 2009. Behavior of red tree voles (Arborimus longicaudus) based on continuous video monitoring of nests. Northwest Science 83: 262–272. https://doi.org/10.3955/046.083.0309.
Forsman, E. D., J. K. Swingle, D. B. Lesmeister, C. A. Marks-Fife, and M. A. Linnell. 2019. A tale of two voles: A response to Rosenberg 2019. Forest Ecology and Management 451: 117258. https://doi.org/10.1016/j.foreco.2019.03.050.
Gomez, D. M., and R. G. Anthony. 1998. Small mammal abundance in riparian and upland areas of five seral stages in western Oregon. Northwest Science 72: 293–302.
Griffey, V., B. Kellogg, R. Haugo, and V. Kane. 2020. Ownership patterns drive multi-scale forest structure patterns across a forested region in southern coastal Oregon, USA. Forests 12: 47. https://doi.org/10.3390/f12010047.
Guerra, R. F., and C. Ades. 2002. An analysis of travel costs on transport of load and nest building in golden hamster. Behavioural Processes 57: 7–28. https://doi.org/10.1016/S0376-6357(01)00207-8.
Hansell, M. H. 2000. Bird nests and construction behaviour. Cambridge, UK: Cambridge University Press. https://doi.org/10.1017/CBO9781139106788.
Ibarra, J. T., K. L. Cockle, T. A. Altamirano, Y. Van Der Hoek, S. W. Simard, C. Bonacic, and K. Martin. 2020. Nurturing resilient forest biodiversity: Nest webs as complex adaptive systems. Ecology and Society 25: art27. https://doi.org/10.5751/ES-11590-250227.
Ingold, D. J. 1994. Influence of nest-site competition between European starlings and woodpeckers. Wilson Bulletin 106: 227–241.
Jackman, R. E., W. G. Hunt, J. M. Jenkins, and P. J. Detrich. 1999. Prey of nesting bald eagles in northern California. Journal of Raptor Research 33: 87–96.
Jiménez-Franco, M. V., J. Martínez-Fernández, J. E. Martínez, I. Pagán, J. F. Calvo, and M. A. Esteve. 2018. Nest sites as a key resource for population persistence: A case study modelling nest occupancy under forestry practices. PLOS ONE 13: e0205404. https://doi.org/10.1371/journal.pone.0205404.
Jones, P. D., B. Hanberry, and S. Demarais. 2009. Stand-level wildlife habitat features and biodiversity in southern pine forests: A review. Journal of Forestry 107: 398–404. https://doi.org/10.1093/jof/107.8.398.
Kempenaers, B., and A. A. Dhondt. 1991. Competition between blue and great tit for roosting sites in winter: An aviary experiment. Ornis Scandinavica 22: 73–75. https://doi.org/10.2307/3676624.
Larsen, K. W., and S. Boutin. 1994. Movements, survival, and settlement of red squirrel (Tamiasciurus hudsonicus) offspring. Ecology 75: 214–223. https://doi.org/10.2307/1939395.
Lesmeister, D., and J. Swingle. 2017. Field guide to red tree vole nests. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station.
Linnell, M. A., R. J. Davis, D. B. Lesmeister, and J. K. Swingle. 2017. Conservation and relative habitat suitability for an arboreal mammal associated with old forest. Forest Ecology and Management 402: 1–11. https://doi.org/10.1016/j.foreco.2017.07.004.
Linnell, M. A., and D. B. Lesmeister. 2019. Landscape connectivity and conservation prioritization for an old forest species with limited vagility. Animal Conservation 22: 568–578. https://doi.org/10.1111/acv.12496.
Linnell, M. A., and D. B. Lesmeister. 2020. Predator–prey interactions in the canopy. Ecology and Evolution 10: 8610–8622. https://doi.org/10.1002/ece3.6518.
Linnell, M. A., D. B. Lesmeister, J. D. Bailey, E. D. Forsman, and J. K. Swingle. 2018. Response of arboreal rodents to increased availability of nest substrates in young forests. Journal of Mammalogy 99: 1174–1182. https://doi.org/10.1093/jmammal/gyy111.
Linnell, M. A., D. B. Lesmeister, Z. Yang, and R. J. Davis. 2023. Timber harvest and wildfires drive long-term habitat dynamics for an arboreal rodent. Biological Conservation 279: 109779. https://doi.org/10.1016/j.biocon.2022.109779.
Mainwaring, M. C., and I. R. Hartley. 2013. The energetic costs of nest building in birds. Avian Biology Research 6: 12–17. https://doi.org/10.3184/175815512X13528994072997.
Marks-Fife, C. A. 2016. Estimation of population age structure, detection probability, and density of red tree voles in Oregon. MSc thesis. Oregon State University. Corvallis, OR.
Maser, C. 1965. Life histories and ecology of Phenacomys albipes, Phenacomys longicaudus, Phenacomys silvicola. MSc thesis. Oregon State University. Corvallis, OR.
Maser, C. 1998. Mammals of the Pacific Northwest: From the coast to the high Cascades. Corvallis, OR: Oregon State University Press.
Maser, C., B. Mate, J. Franklin, and C. T. Dyrness. 1981. Natural history of Oregon coast mammals. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Forest and Range Experiment Station. https://doi.org/10.2737/PNW-GTR-133.
Michel, A. K., and S. Winter. 2009. Tree microhabitat structures as indicators of biodiversity in Douglas-fir forests of different stand ages and management histories in the Pacific Northwest, U.S.A. Forest Ecology and Management 257: 1453–1464. https://doi.org/10.1016/j.foreco.2008.11.027.
Millar, J. S. 2007. Nest mortality in small mammals. Écoscience 14: 286–291. https://doi.org/10.2980/1195-6860(2007)14[286:NMISM]2.0.CO;2.
Moffett, M. W. 2000. What’s “up”? A critical look at the basic terms of canopy biology. Biotropica 32: 569–596. https://doi.org/10.1111/j.1744-7429.2000.tb00506.x.
Nyland, R. D., L. S. Kenefic, K. K. Bohn, and S. L. Stout. 2016. Silviculture: Concepts and applications. 3rd ed. Long Grove, IL: Waveland Press.
Old-Growth Definition Task Group. 1986. Interim definitions for old-growth Douglas-fir and mixed-conifer forests in the Pacific Northwest and California. Research Note PNW-447. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station.
Oregon Department of Fish and Wildlife. 2016. Oregon Conservation Strategy. Salem, OR: Oregon Department of Fish and Wildlife.
Parker, G. G. 1995. Structure and microclimate of forest canopies. In Forest canopies, edited by M. D. Lowman and N. M. Nadkarni, 73–106. San Diego, CA: Academic Press. https://doi.org/10.1016/B978-012457651-2/50010-0.
Pierluissi, S., and S. King. 2008. Relative nest density, nest success, and site occupancy of king rails in southwestern Louisiana rice fields. Waterbirds 31: 530–540. https://doi.org/10.1675/1524-4695-31.4.530.
Price, A. L., J. S. Mowdy, J. K. Swingle, and E. D. Forsman. 2015. Distribution and abundance of tree voles in the northern Coast Ranges of Oregon. Northwestern Naturalist 96: 37–49. https://doi.org/10.1898/NWN14-04.1.
Rosenberg, D. K. 2019. A tale of two voles: The challenge of the commonness–rarity continuum in conservation planning. Forest Ecology and Management 434: 165–171. https://doi.org/10.1016/j.foreco.2018.12.010.
Rosenberg, D. K., R. J. Davis, K. J. Van Norman, J. R. Dunk, E. D. Forsman, and R. D. Huff. 2016. Patterns of red tree vole distribution and habitat suitability: Implications for surveys and conservation planning. Ecosphere 7: e01630. https://doi.org/10.1002/ecs2.1630.
Ruggiero, L. F., K. B. Aubry, and M. H. Brookes. 1991. Wildlife and vegetation of unmanaged Douglas-fir forests. Gen. Tech. Rep. PNW-GTR-285. Portland, OR: U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station. https://doi.org/10.2737/PNW-GTR-285.
Swingle, J. K., and E. D. Forsman. 2009. Home range areas and activity patterns of red tree voles (Arborimus longicaudus) in western Oregon. Northwest Science 83: 273–286. https://doi.org/10.3955/046.083.0310.
Swingle, J. K. 2005. Daily activity patterns, survival, and movements of red tree voles (Arborimus longicaudus) in western Oregon. MSc thesis, Corvallis, OR: Oregon State University.
Thompson, J. L., and L. V. Diller. 2002. Relative abundance, nest site characteristics, and nest dynamics of Sonoma tree voles on managed timberlands in coastal northwest California. Northwestern Naturalist 83: 91–100. https://doi.org/10.2307/3536607.
U.S. Department of Agriculture and U.S. Department of the Interior. 1994. Final supplemental environmental impact statement on management of habitat for late-successional and old-growth forest related species within the range of the northern spotted owl. Portland, OR: U.S. Department of Agriculture, Forest Service, and U.S. Department of the Interior, Bureau of Land Management.
U.S. Fish and Wildlife Service. 2019. Species status assessment: North Oregon Coast population of the red tree vole (Arborimus longicaudus). Portland, OR: U.S. Fish and Wildlife Service.
U.S. Fish and Wildlife Service. 2022. Endangered and threatened wildlife and plants; Withdrawal of the not-warranted finding for endangered or threatened status for the North Oregon Coast distinct population segment of red tree vole. Federal Register 87: 63472–63473.
U.S. Fish and Wildlife Service. 2023. Species status assessment report version 2.0: North Oregon Coast distinct population segment of the red tree vole (Arborimus longicaudus). Portland, OR: U.S. Fish and Wildlife Service.
Van Pelt, R. 2007. Identifying mature and old forests in western Washington. Olympia, WA: Washington State Department of Natural Resources.
Warde, W. D., and J. W. Petranka. 1981. A correction factor table for missing point-center quarter data. Ecology 62: 491–494. https://doi.org/10.2307/1936723.
White, G. C., and R. A. Garrott. 1990. Design of radio-tracking studies. In Analysis of wildlife radio-tracking data, 13–25. San Diego, CA: Academic Press. https://doi.org/10.1016/B978-0-08-092657-5.50005-7.
Open Access
Peer-Reviewed
Creative Commons
Accepted: 20 January 2026
Published: 27 March 2026
Funding was provided by the National Council for Air and Stream Improvement Inc., Oregon State University through the Fish and Wildlife Habitat in Managed Forests Research Program, Oregon Department of Forestry, Weyerhaeuser, US Fish and Wildlife Service and National Alliance of Forest Owner’s ‘Wildlife Conservation Initiative’, and the Oregon Wildlife Foundation.
Conflicts of Interest:
The authors declares no conflicts of interest.