An initial assessment of risk to pollinators from mosquito control in residential settings
1 The Xerces Society for Invertebrate Conservation, Portland, Oregon
2 Synergistic Pesticide Laboratory, Portland, OR
3 Program in Ecology, Evolution and Conservation Biology, Department of Biology, University of Nevada, Reno, Nevada
AA: https://orcid.org/0000-0002-0332-9827
RM: https://orcid.org/0000-0003-0144-5928
MLF: https://orcid.org/0000-0003-2765-4779

Abstract photo. Adult mosquito. Credit: Katja Schulz/Flickr, CC BY 2.0
Abstract
Sprays to kill adult mosquitoes are a source of insecticide exposure to beneficial insects, including pollinators,but the risks to pollinators are not well quantified. In this study, silicone bands were deployed to capture drift from hired home mosquito sprays and from mosquito control district (MCD) sprays. We found an average insecticide load of 1563.6 ng/g per band in hired spray yards, 28.6 ng/g in neighboring yards, and 7.3 ng/g in yards exposed to MCD sprays. We calculated honey bee “risk quotients” (an RQ of 1 = the LD50 for honey bees), and found concerning potential for risk to pollinators, with an average pyrethroid insecticide RQ of 6.6, 0.12, and 0.03 for yards with hired sprays, neighbor drift, and MCD sprays, respectively. Even though the latter two categories had lower risk quotients, there was considerable variation and some individual yards showed elevated levels with the potential to harm pollinators. We also calculated risk quotients for specific pyrethroids and three species of butterflies, and found levels of risk tied to moderate to high levels of risk in many yards. Our results suggest that, on a site by site basis, residential mosquito sprays, in particular barrier sprays performed by private companies, likely pose a significant risk to pollinators.
Keywords: barrier spray, insecticides, mosquito control, pesticide drift, pollinators, urban habitat
Introduction
Across the United States, many wild bee species have declined in abundance and their ranges have contracted (Colla and Packer, 2008, Cameron et al., 2011, Bartomeus et al., 2013, Jacobson et al., 2018, Richardson et al., 2019, Guzman et al., 2024). Butterflies in North America have similarly undergone severe reductions in population density across diverse habitats and landscapes (Wepprich et al., 2019, Forister et al., 2021, Edwards et al., 2025, Leuenberger et al., 2025). These declines have been attributed to multiple stressors, including habitat loss, climate change, disease, and pesticide use (Raven and Wagner, 2021, Wagner et al., 2021, Guzman et al., 2024). Here, we add to our understanding of stressors by investigating pesticide contamination associated with adult mosquito treatments in residential areas in three US states.
Although residential areas are associated with loss of natural habitat, pesticide use, and elevated rates of warming due to the urban heat island effect, some of these landscapes have shown the ability to harbor diverse and abundant pollinator communities (Fetridge et al., 2008, Lowenstein et al., 2014, Baldock et al., 2015, Theodoreau et al., 2020, Wenzel et al., 2020). Site level management, including planting a diversity of flowering plants, can help to counter the landscape-wide impact of urbanization, and home gardeners have contributed to this effort to increase habitat for insects.
However, even when habitat is present in urban areas, pollinators and other beneficial insects can still be negatively impacted by pesticide use. Background contamination can be widespread, having been detected in urban greenspaces like natural areas, community gardens and yards (Halsch et al., 2020, Siviter et al., 2023, Dittemore et al., 2025). The risks that insecticides pose to pollinators are varied and are dependent on factors including the species of insect, the compounds used, and the application method. Some insecticide applications can expose pollinators to lethal levels, but even low-level exposures can cause negative effects such as altered offspring sex ratios (Sandrock et al., 2013), changes in learning (Decourtye et al., 2004), impaired foraging (Gill and Raine, 2014), and difficulty navigating (Fischer et al., 2014). Though these effects might not kill the individual, they can have negative population-level consequences (Raine and Rundlöf, 2024).
Pesticide exposure in residential landscapes can come from many sources, including sprays intended to kill adult nuisance or disease-carrying mosquitoes, a practice known as “adulticiding.” These applications are generally performed by either hired pest control services who spray individual yards, or by mosquito control districts that coordinate mosquito management across communities. Private mosquito spray companies most often rely on pyrethroid insecticides, which kill insects by interfering with the sodium ion channels in their nervous systems (Narahashi et al., 1992, Katsuda, 2011, Jeran et al., 2020). These applications are called “barrier sprays”, using relatively large droplets applied to vegetation in the yard via a backpack sprayer (Stoops et al., 2019). The applications are designed to kill mosquitoes roosting in the vegetation and provide a repellent barrier (Cilek, 2008, Fulcher et al., 2015, Stoops et al., 2019). This type of treatment is purported to last for several weeks, and services often return every three weeks (see for example, the private companies Mosquito Joe and Mosquito Authority).
In contrast to private mosquito control operations, vector or mosquito control districts (MCDs) are governmental or quasi-governmental organizations charged with controlling disease vectoring organisms. They tend to use “ultra-low volume” (ULV) sprays to target adult mosquitoes, often called “fogging” because the insecticides are aerosolized into a fine mist or fog. ULV applications are made via a ground-truck mounted sprayer or aircraft, and produce very small droplet sizes (5-25 um) (Bonds, 2012, Stoops et al., 2019) to facilitate pesticide drift across the landscape. Depending on droplet size, these sprays can travel over 100m (Bonds, 2012). Like private companies, control districts often spray pyrethroids, though organophosphates are also used (Tai et al., 2024). Different control districts have different practices: some spray on a set schedule, while others monitor disease prevalence and mosquito populations and only spray when thresholds are met. Generally, MCDs spray throughout entire neighborhoods or communities, though some districts will respond to individual resident complaints or otherwise target treatments.
Despite these mosquito treatments being used across wide parts of the United States, relatively little is known about the risks these sprays pose to pollinators, and this is true in particular of residential barrier sprays. Previous research has found that monarch caterpillars fed leaves sprayed by a permethrin mosquito barrier treatment had much lower survival rates than control caterpillars for up to 21 days after an application (Oberhauser et al., 2006). These sprays may also pose a risk to honey bees (Qualls et al., 2022). Even less is known about the extent to which spray applications drift from one residential yard to another, or how such drift might impact insect communities.
Comparatively more research exists on the non-target impacts of mosquito control district sprays. While some studies have found minimal impacts to bees and butterflies (Boyce et al., 2007, Pokhrel et al., 2018), others have found both lethal (Oberhauser et al., 2009, Rinkevich et al., 2017) and sublethal impacts from MCD sprays, including the development of smaller adult monarchs (Oberhauser et al., 2009), and mass insect kills have been observed after applications (Domonoske, 2016, Nelson, 2020). A probabilistic risk assessment suggested permethrin and naled MCD sprays posed risks to butterflies in Florida (Hoang and Rand, 2015).
The purpose of this study is to investigate insecticide residues in yards as a result of mosquito spray applications under multiple circumstances. Specifically, we ask if insecticide loads and the number of accumulated compounds differ when comparing yards that were directly sprayed by private mosquito control services as compared to yards neighboring those directly sprayed yards. We also characterize contamination associated with mosquito control district applications. Finally, we use available literature to quantify the risks posed to insects, focusing on bees and butterflies.
Methods and Materials
Volunteer recruitment and site selection
We recruited volunteers in three states: Georgia, Iowa, and Massachusetts. In Georgia, all the volunteers were located in Decatur, recruited by the chair of the Xerces Society’s Bee City USA (BCUSA) program via BCUSA and native plant society email lists. In Iowa, Good Neighbor Iowa recruited volunteers from their Local Champions program, and a Xerces employee with connections to the Massachusetts Pollinator Network program helped recruit volunteers across eastern Massachusetts.
We screened potential volunteers via Google Forms (Mountain View, CA), a web-based survey tool, to determine the suitability of yards for inclusion in our study. We asked whether the volunteers themselves hire companies to spray for mosquitoes, whether their neighbors hire companies to spray for mosquitoes, or whether the local mosquito control district sprays for mosquitoes. If they answered yes to any of these, we asked what months these sprays occur, if they knew the estimated spraying schedule, and their level of confidence in knowing whether a spray event occurred. Respondents who answered affirmatively to these questions and expressed high confidence were included in our initial pool of volunteers, and we ended up with 19 volunteers across the three states.
Sampling
To capture deposition from mosquito adulticide sprays, we used silicone bands as sentinel monitors to passively capture drift. Silicone bands have been used to measure the presence of pesticides in environmental studies, as silicone is inert and known to absorb a variety of chemicals from the air (O’Connell et al., 2014). This makes silicone bands uniquely suited for measuring deposition from residential mosquito sprays.
Silicone bands (www.24hourwristbands.com, 1.2 cm wide) were cleaned and prepared (O’Connell et al., 2014) by Synergistic Pesticide Laboratory, LLC (Portland, OR), and wrapped in aluminum foil. We mailed the bands to volunteers along with written instructions and supplies: plastic bags, and nitrile gloves for handling, and 54cm long sections of wire with hooks bent onto the end (Figure 1) to hang the bands. Volunteers placed these wires into the ground so that the bands hung ~35cm above the ground. Volunteers deployed bands a few hours to a day before a spray event was scheduled to occur.
We measured three different types of spray events: a hired service in the participant’s yard, drift from a spray hired by a neighbor, or a mosquito control district spray. Volunteers placed two bands on the edge of the property line where the pesticide drift was likely to come from (i.e., the property line with a neighbor who sprayed, or the edge of the road for an MCD spray), and a third in between these bands set 6.1 meters (20 feet) back into the yard. We instructed volunteers who hired spray services to place two bands along the outside edges of their property, with one in the center of the yard.
Two of the volunteers had no known neighbor or MCD sprays, and they placed the bands in their yards for seven days as non-sprayed controls. One participant who hired a spray service placed bands in both their own yard and in the adjacent neighbor’s yard, to capture the spray event from two sites. Two other participants placed and collected bands for multiple distinct spray events on different dates.
One band was sent to a volunteer in each state as “trip check” controls to ensure there was no pesticide contamination in transport or handling. The volunteers handled these control bands like the sample bands, but instead of placing them in the field they immediately wrapped them in foil and placed them in their freezers.
Volunteers recorded the dates they deployed and retrieved the bands. Bands were generally out in the field for 24 to 48 hours with the goal of capturing only the discrete spray event and associated drift. Volunteers then collected the bands, wrapped them in aluminum foil, and stored them in plastic bags in their freezers. Once sampling was complete in each state, a point-person collected all the bands in that state, packed them in a cooler with ice packs, and shipped them overnight to the lab. They were stored at < -15.0° C for approximately six months before chemical analysis.
Chemical analysis
The lab extracted compounds from the silicone bands twice using sonication with 50ml dichloromethane, followed by concentration via rotary evaporator and nitrogen. Extracts were reconstituted in ACN and analyzed for 293 compounds using both liquid chromatography mass spectrometry (LC/MS/MS) and gas chromatography mass spectrometry (GC/MS/MS). A full list of the compounds screened and analytical limits of detection are included in the Appendix S1. The lab performed quality controls for every batch of 20 samples on cleaned, non-deployed silicone bands (n=8). These included matrix spikes and matrix spike duplicates, which were samples of known pesticide amounts to validate compound recoveries, as well as blanks, which were clean bands to confirm an absence of contamination within the analytical system. All method blanks were non-detect.
GC/MS/MS analysis was performed using a Thermo TSQ 8000 Evo mass spectrometer with a Trace 1310 GC and AS 1310 Autosampler. Injections were made onto a PTV liner (Restek Topaz liner, baffled; PN 23438) using a Zebron guard column (Phenomenex; PN 7AG-G000-00-GZK) and Zebron ZB-5MS plus column (Phenomenex 30m x 0.25 (0.25); PN 7HG-G030-11). The column temperature ramp started at 60°C and increased to 200°C at a rate of 20°C/minute, held for three minutes, then increased to 310°C at a rate of 7°C/minute for a total run time of 30 minutes.
LC/MS/MS analysis was performed using a Thermo TSQ Endura with Vanquish binary pump and autosampler. A SecurityGuard ULTRA Cartridges guard column (Phenomenex UHPLC Biphenyl 2.1 µm; PN AJ0-9209) and Kinetix Biphenyl column (Phenomenex 100 x 2.1 (2.6 µm); PN 00-4622-AN) were used with a flow rate of 0.3 mL/minute. The gradient program began with 100% of an aqueous solution (A) containing 0.05% formic acid, 0.01% ammonium hydroxide, and 2% MeOH. This mobile-phase composition was held for 1.5 minutes and ramped to 25% of an MeOH solution (B) containing 0.05% formic acid, 0.01% ammonium hydroxide and held from 5 to 6.5 minutes before ramping up to 80% (B), held from 19-21.5 minutes and finally ramped to 100% (B) for a total run time of 30 minutes.
For both GC and LC analyses, one qualitative transition and at least one qualifier transition were monitored for each analyte. Retention times and ion ratios of quan and qual ions were determined from analytical standards provided by ChemService, Sigma Aldrich, AccuStandard, and LGC. All analyses were calibrated with a minimum of a five-point calibration curve, and samples were bracketed with continuing calibration verification checks.
Calculating a risk quotient
Honey bees
To estimate the risk posed to bees directly sprayed by residential mosquito sprays, we calculated a cumulative pyrethroid Risk Quotient (RQ) based on the contact doses required to kill 50% of a honey bee (Apis mellifera) test population (LD50). The Risk Quotient method is used by various regulatory bodies to assess risk by looking at toxicity and exposure (EPA, 2007), and is broadly calculated as RQ = Environmental Exposure ÷ Toxicity. We used honey bees as a representative organism because honey bee LD50s are known for most compounds due to their use in regulatory testing. There is, unfortunately, no standard method to directly relate the residues found on silicone bands to honey bee risk. However, Ward et al. (2022) note that pesticide concentrations from silicone bands are more dependent on surface area than band mass. To calculate exposure by surface area, we measured the dimensions of the bands to get a total band surface area (55.73 cm2) and divided the per-band residue values by that surface area to get ng/cm2 value.
To estimate how much spray a bee might receive, we referenced Poquet et al. (2014), who found that on average 1.05 cm2 of surface area is covered when a honey bee is sprayed directly by a Potter spray tower. Multiplying our ng/cm2 values by 1.05 cm2 allowed us to estimate the amount of insecticide a honey bee would encounter if sprayed.
We found acute contact LD50 values for honey bees for each insecticide detected from the University of Hertfordshire Pesticide Properties Database (Lewis et al., 2016) and divided the per-bee insecticide exposure values from our data by their respective LD50s to get a Risk Quotient for each insecticide. In this case, a RQ of 1.0 would mean the amount detected was at the honey bee LD50 level for that pesticide. Since pyrethroids have the same mode of action, we considered their effects to be additive and summed the RQs for all of the detected pyrethroids, resulting in a cumulative pyrethroid RQ (Stoner et al., 2019). This represents an index of the total risk to honey bees from pyrethroid insecticides in each sample. The cumulative RQ does two things: it allows for the total risk to be assessed at each site, and allows for better comparison between sites.
Using the same method, we also calculated honey bee risk quotients for the two neonicotinoid insecticides detected (thiamethoxam and imidacloprid) and summed them, since they too have the same mode of action, producing a cumulative neonicotinoid RQ. We are uncertain if these neonicotinoids were used in mosquito control or if the residues came from a different application; still, we wanted to account for their presence on the bands.
We classified the cumulative RQ values to five risk levels: very high risk ( RQ ≥ 10), high risk (RQ < 10 and ≥ 1), medium risk (RQ < 1 and ≥ 0.1), low risk (RQ < 0.1 and ≥ 0.01) and negligible risk (RQ < 0.01 and ≥ 0.001) (Sanchez Bayo et al., 2002, Iturburu et al., 2019). For reference, in the U.S. Environmental Protection Agency (EPA)’s tiered approach to assessing pesticide risk to bees, 0.4 is the Tier 1 RQ for the acute contact exposure risk to honey bees (US EPA, 2014).
Monarch butterflies
Unfortunately, it is difficult to assess possible risk to most insects since LD50s for most pesticides are not available for the vast majority of invertebrate species, let alone threatened or endangered species. However, a bifenthrin contact LD50 has been reported for monarch butterflies (Danaus plexippus), and we used this value to investigate risk for this species that was recently proposed for listing under the Endangered Species Act (Federal Register, 2024). Bifenthrin has an LD50 of 0.44 ug/larva for fifth-instar monarch butterflies as calculated from Krueger et al. (2021). Krishnan et al. (2020) estimated the average surface area of fifth-instar monarch larvae to be 7.1 ± 1.3 cm2, by assuming them to be cylindrical and measuring their height and length. They estimated one half of the surface area of a larva would be contacted by a pesticide spray application.
To calculate the bifenthrin Risk Quotient for fifth-instar monarch larvae, we used the same method as discussed above for honey bees, but used 0.44 ug/larva as the LD50 value and 3.55 cm (one half of the surface area of a larva) as the surface area value. The resulting values only represent the bifenthrin RQ, not a cumulative pyrethroid RQ, since there are no published LD50s for the other compounds.
Other butterfly species
For both the common buckeye (Junonia coenia) and painted lady (Vanessa cardui) butterflies, published permethrin LD50s are available for fifth-instar larvae (Hoang et al., 2011). However, these values were not accompanied by estimates of larval surface area or dimensions. To estimate the surface area of these species, we found published fifth-instar lengths for both: 38mm for the common buckeye and 32mm for the painted lady (James et al., 2011). We could not find published larval widths, so we used a digital ruler to obtain the length and width of larvae in representative images (five common buckeye, three painted lady: bugguide.net). We then calculated an average ratio of length to width for each caterpillar species using these images. We used these ratios and the published average lengths to calculate average widths: 4.6mm and 4.4mm for the common buckeye and painted lady, respectively.
Using these dimensions, we used the same methods described above for monarchs to calculate permethrin RQs for these species. We assumed the caterpillars to be cylinders and that one half of their surface area would be sprayed. We could not calculate a cumulative pyrethroid RQ since we had no LD50s for other compounds for these species.
Statistical analyses
We used linear, mixed effects models to quantify the effect of spray type (hired spray vs. drift from spray in a neighboring property) and position (band on the edge or middle of yard) on pyrethroid load (the total amount deposited on a band) and pesticide richness (the number of unique compounds detected), while accounting for individual yard as a random effect (allowing for random intercepts). For the model of pesticide richness, we also included “state” as a random effect. For the other model (of pyrethroid load), inclusion of “state” produced a singular model fit and was thus not retained, although results for main factors of interest with and without “state” were qualitatively identical.
We focused on pyrethroids in a model of pesticide load because pyrethroids are the dominant class of pesticides discovered, and we analyzed load rather than RQ (discussed below), because the latter produced highly non-normal residual variance. Also, we excluded MCD applications as a spray type from models because the MCD sprays were confounded with state, with all but one of the yards subject to MCD sprays in Iowa.
Pyrethroid load was natural log transformed (after adding 1 to account for a small number of cases with no detected pesticides) and modeled with a Gaussian error, while the model of compound richness used a Poisson distribution and log-link function. All analyses were conducted using R (v.4.2.3), and models were run using the glmer and lmer functions in the lme4 package (Bates et al., 2015). Coefficients of determination (R2) were calculated from model outputs using the r.squaredGLMM function in the MuMIn package (Bartoń, 2025); residuals were simulated and visualized using the DHARMa package (Hartig, 2024). A modified version of the pheatmap function from the pheatmap package (Kolde and Kolde, 2015) was used to create a heatmap of concentrations across all samples and compounds; the modified version is included with archived code (see “Data Availability”).
Results
Compounds detected
A total of 21 compounds were detected in our study. Of these, 12 were insecticides, eight were fungicides, and one was a synergist designed to increase the toxicity of certain insecticides (Figure 2). Pyrethroid insecticides were detected on 56 out of 70 non-control bands, with bifenthrin detected on 53 out of 70 bands. A total of five pyrethroid compounds and one pyrethroid derivative were detected. Additionally, we found two neonicotinoids, three insect growth regulators (IGR), and carbaryl (a carbamate). All the fungicide detections (azoxystrobin, carbendazim, flutriafol, imazalil, propiconazole, pydiflumetofen, pyraclostrobin, and trifloxystrobin) were in Iowa. Since they are unlikely to be related to the mosquito spray events, we excluded these from our analyses.
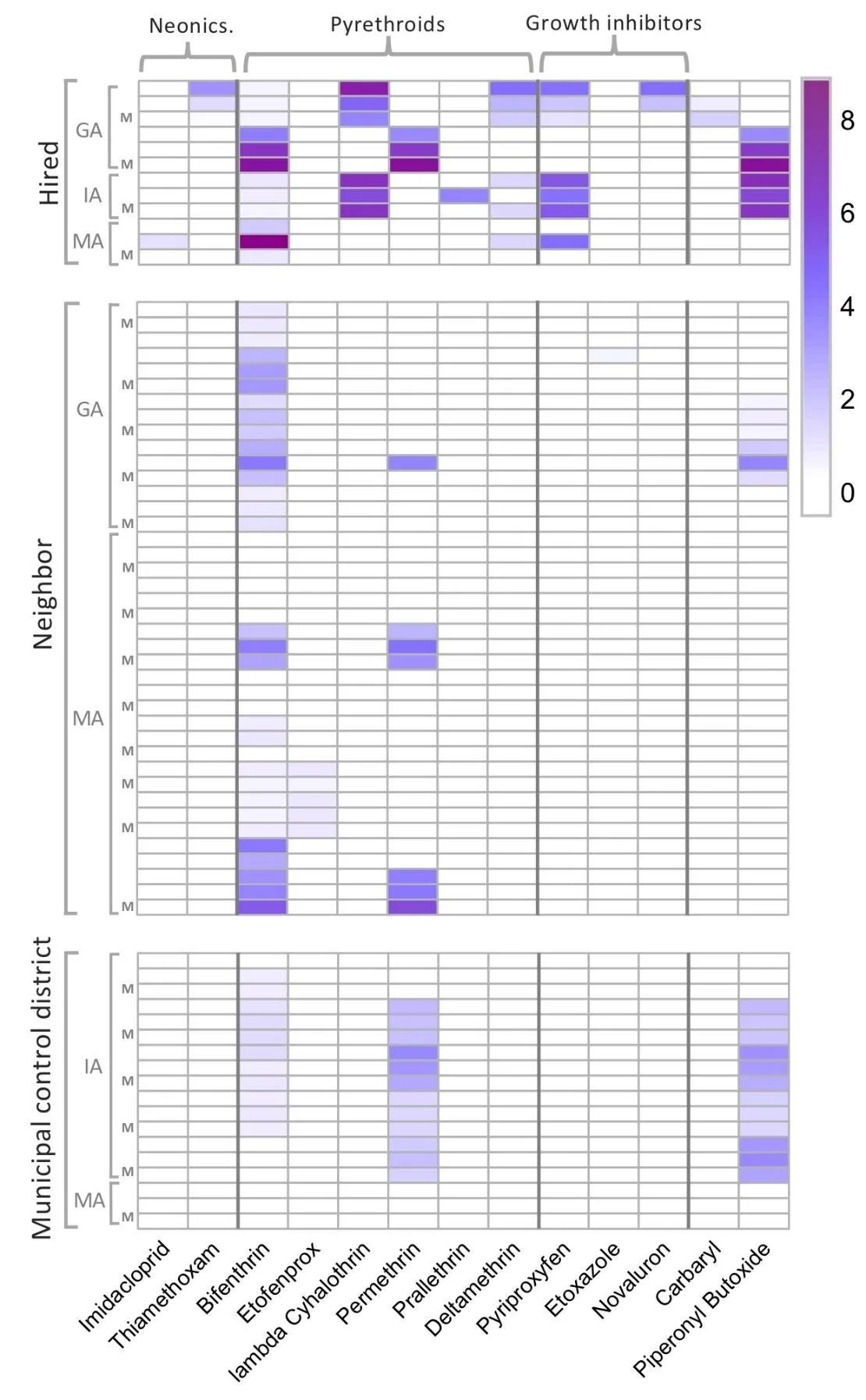
Figure 2. Heatmap summarizing concentrations of specific compounds in ng/g (ppb) on a natural log scale, with the following organization: columns are individual compounds (with compound names given along the bottom) and are grouped into classes (see labels across the top); rows represent individual silicone bands and are grouped by spray type (hired, neighbor or MCD), then by state, and then clustered by yard. Individual yards are not labelled, but for each yard there are edge samples followed by a sample in the middle of the yard marked with 'M' along the left margin.
A model of pesticide richness as a function of spray type (hired or neighbor) and sampling position, including state and yard as random effects, detected a strong effect of spray type: the estimate of the spray type effect for the neighbor treatment (comparing against hired as the reference) was -1.15 (std. error = 0.28; z = -4.09, P < 0.001) in a model that explained a third or more of the variance (marginal R2 = 0.31 and conditional R2 including random effects = 0.41). On average, 4.1 ± 0.6 (std. error) distinct compounds were found in bands from yards that hired a mosquito treatment, as compared to 1.2 ± 0.1 compounds found on average in neighboring sprayed yards (Figure 3). The effect of middle position, in contrast, was -0.11, compared to edge as the reference (std error = 0.22, z = -0.50, P = 0.61) (Figure 3).
Pyrethroids were the dominant insecticide class of mosquito spray treatments overall, and certain pyrethroids were detected more often in private home sprays (hired and neighboring yards) compared to MCD sprays (Figure 2). Deltamethrin, lambda cyhalothrin, and prallethrin were found only in privately sprayed yards. Permethrin and bifenthrin were detected frequently in yards sprayed by private services and by mosquito control districts (Figure 2). The synergist piperonyl butoxide was found in both private and MCD sprays, but was slightly more common in MCD sprays, being found in all but two yards with detectable pesticide residues sprayed by an MCD.
The “trip check” control bands had no compounds detected on them, indicating no contamination in handling or transport. The control bands placed in unsprayed yards in Georgia and Iowa had no detections, except for one band in Iowa that had small amounts of permethrin (3.21 ng/g) and piperonyl butoxide (1.31 ng/g).
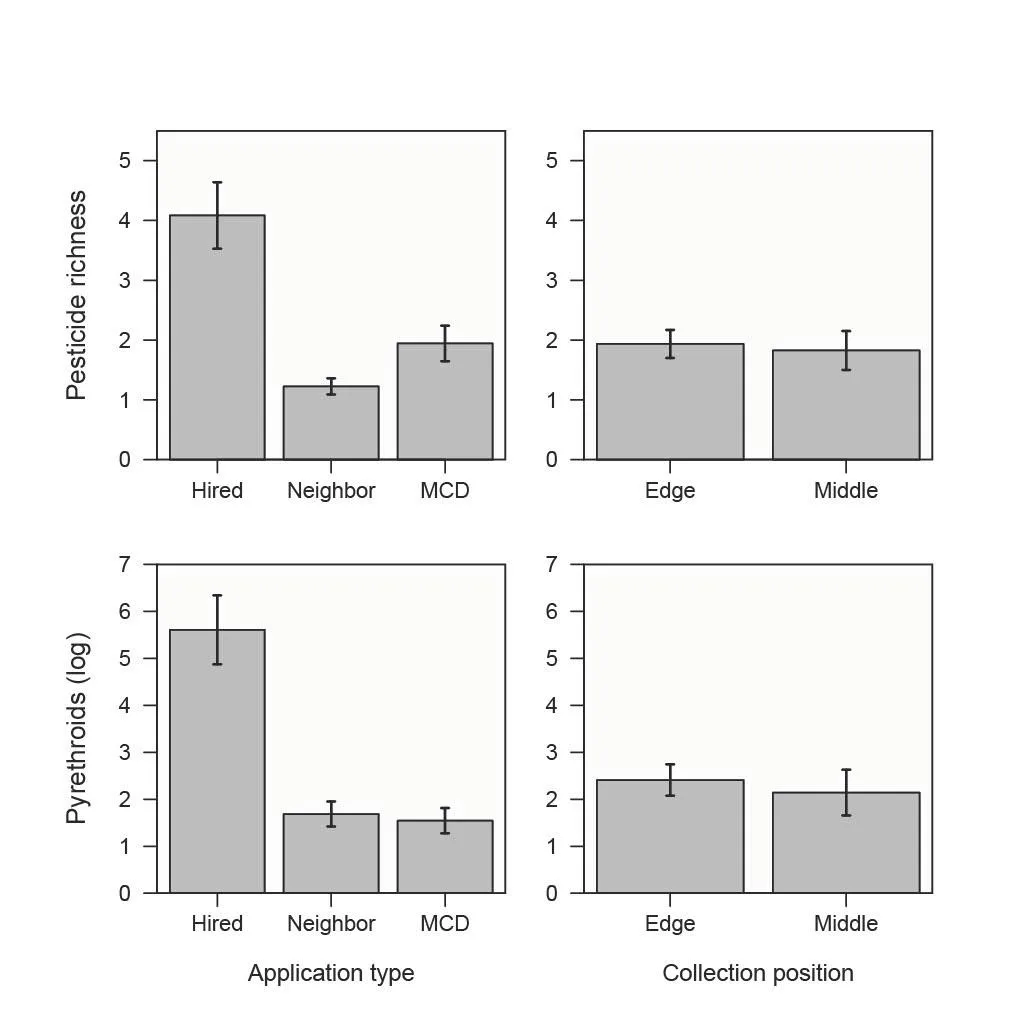
Figure 3. Summaries of pesticide richness (top panels) and pesticide load (on a natural log scale, bottom panels), organized by application type (left column) and collection position (right column). In all panels, bars show means and standard errors around means. MCD sprays are shown here for comparison but were excluded from statistical models because they were confounded with state (mostly from Iowa).
Insecticide load
Considering insecticides detected (removing piperonyl butoxide), bands placed in hired yards contained, on average, 1563.6 ± 665.7 ng/g as compared to 28.6 ± 12.7 ng/g in neighbor yards, and the average per band contamination associated with mosquito control district sprays was 7.3 ± 2.3 ng/g. Focusing on pyrethroids as the dominant group of insecticides, we found a similar pattern: 1499.4 ± 663.9 ng/g in hired yards compared to neighbor yards where bands collected drift at an average rate of 28.6 ± 12.7 ng/g per band, and 7.3 ± 2.2 ng/g in yards sprayed by mosquito control districts. We used a linear mixed model to estimate effects on pyrethroid contamination (excluding mosquito control district sprays, as mentioned above). The estimated effect of spray type for pyrethroid load, comparing neighbor spray yards against the reference of hired yards, was -3.72 (std. error = 0.88, t = -4.22, P = 0.0012). The marginal variance explained by the model was 0.39, and conditional R2 with the random effect of years was 0.60. As with the previous model, the effect of middle yard position compared to edge was weak but negative (-0.26, std. error = 0.48, t = -0.54, P = 0.59), which raises the possibility of fine scale spatial effects that could be examined in future studies with greater sample size.
Risk quotients
Cumulative pyrethroids and honey bees
Of the 12 samples from the four yards directly sprayed by barrier treatments, ten had residues found at or above a medium risk level. Six had pyrethroid levels above the honey bee cumulative pyrethroid LD50 (Table 1), with an average pyrethroid Risk Quotient of 6.6 ± 3.2, (over six times the honey bee LD50). All four of these yards had at least one band that exceeded the honey bee pyrethroid LD50, with one sample 24 times higher and another over 34 times higher. Yards that experienced drift from a neighboring application had an average pyrethroid RQ of 0.12 (± 0.05), and nine of the 40 samples (23%) at five out of 14 sites had residues at medium or high risk levels). One sample had residues over the honey bee cumulative pyrethroid LD50.

Table 1. Number of samples detected at various risk quotient (RQ) levels, with the right-hand column displaying the maximum RQ value calculated. The top section shows cumulative pyrethroid and neonicotinoid RQs based on honey bee LD50 values, and the bottom section displays specific pyrethroid insecticide RQs calculated for three butterfly species for which these toxicity data exist.
Mosquito control district spray events resulted in the lowest pyrethroid RQ values (0.03 ± 0.01). One band had pyrethroid residues at the medium risk level (0.13). The remaining 17 samples across five sites all had pyrethroid RQ values below 0.10.
Neonicotinoids and honey bees
Neonicotinoid insecticides (imidacloprid and thiamethoxam) were detected in two yards, on four bands total (Figure 2; and note that one of the four values was a very low concentration). After calculating additive neonicotinoid Risk Quotients for honey bees, one of these samples had contamination at a medium risk level (0.10) (Table 1). Both of these yards were sprayed directly by hired services.
Bifenthrin and monarchs
All samples (n = 12) from the yards receiving barrier treatments (n = 4) had bifenthrin residue. Of these, two samples in two different yards were over the fifth-instar monarch LD50 (one 1.7 times the LD50, and another 4.2 times the LD50) (Table 1). One of the yards receiving drift from a neighboring spray that had detectable bifenthrin residue had a band with a monarch RQ at the medium risk level. None of the MCD sprays had bifenthrin values over the low risk level.
Permethrin and common buckeyes and painted ladies
Only one of the yards receiving a barrier treatment had detectable permethrin residue. Two of the samples in this yard had fifth-instar common buckeye Risk Quotients well above the permethrin LD50 with RQs of 7.9 and 1.7. Across all samples, seven had contamination at or above the medium risk level for common buckeyes. Painted ladies are less susceptible to permethrin, yet one of the samples was over twice the LD50 for fifth-instar painted ladies (2.2), and a second sample was about half the LD50 (0.48).
Discussion
Our results indicate that residential mosquito sprays likely pose a significant risk to bees and other beneficial insects. In particular, yards receiving barrier sprays from private companies had residues up to 34 times the honey bee cumulative pyrethroid LD50. While the level of risk is clearly less in neighboring yards, they still showed concerning residues; the 0.12 RQ average for those yards was within the “medium-risk” classification. Looking closer, average values only tell part of the story, and it is illustrative to examine values on a yard-to-yard basis, since insects will encounter individual surfaces within individual yards rather than the average across yards. Five yards had pyrethroid drift over the medium-risk level, and three separate yards had neighbor drift at levels above the EPA’s honey bee acute-risk level of concern. So, while risk was much lower when compared with yards sprayed directly, the potential for harm from neighbor spray is still present, especially for sensitive or vulnerable species. This suggests that people creating pollinator habitat in urban yards should consider contamination from mosquito sprays and look for mitigation opportunities.
On average, yards sprayed by mosquito control districts had the lowest total pyrethroid loads and lowest pyrethroid risk quotients out of our sampled yards. This is likely because these sprays utilize foggers that aerosolize very small pesticide droplets, and use smaller quantities of active ingredients when compared with home barrier sprays. Only one of the sampled yards registered residues at the moderate risk level. While our findings suggest relatively limited contamination from these applications, it is important to remember the short duration of our study and that we only sampled a small handful of districts that were mostly in one state. Different MCDs have different practices, and use different insecticides and equipment. It is notable that significant mortality events have occurred after MCD sprays. These include the monarch kill after a permethrin spray in Cass County, SD (Nelson, 2020), and honeybee colony loss after an aerial naled spray in Dorchester County, SC (Domonoske, 2016). Many sources of heterogeneity in applications remain unquantified, including variation in active ingredients, different spray equipment, or operator error. We also could not account for the impacts of the synergist piperonyl butoxide, which increases the toxicity of pyrethroids and was found at four out of the six MCD sprayed sites. Furthermore, these sprays often occur across many acres, and in this study we looked only at specific sites. We recognize that there is much yet to be learned about accumulated effects potentially associated with MCD sprays which are often performed on set schedules throughout mosquito season, and cover broad areas.
Mosquito treatments are also likely to adversely impact pollinator species beyond bees. Though pesticide toxicology data for Lepidoptera are scarce, we were able to find several contact LD50 values for three butterflies that provide a glimpse into impacts of mosquito sprays on Lepidoptera. Two hired yards had bifenthrin residues well above the fifth-instar monarch LD50. Though permethrin was found in far fewer yards, there were still samples with permethrin residues above the LD50 for common buckeye and painted lady butterflies. This suggests these sprays can be a source of acute risk for Lepidoptera. Even when pyrethroid residues are below the LD50, there is still risk potential. For example, hemolymph bleeding and spasming of monarch larvae were observed when exposed to bifenthrin levels less than half the LD50 (Krueger et al., 2021).
Residential areas can harbor diverse pollinator communities, including species of conservation concern. For example, surveys have frequently found the federally endangered rusty-patched bumble bee (Bombus affinis) in urban and suburban yards, and the monarch butterfly, a proposed species for listing under the US Endangered Species Act, is also commonly found throughout residential landscapes (Crone and Schultz, 2021). Both of these species are found in the three states where we sampled, underscoring the importance of protecting urban pollinator habitat from pesticides when vulnerable species might be present. In addition, five other bumble bee species of conservation concern are found throughout the states we sampled (IUCN, 2024). Of course, most insect populations are not monitored, so there are potentially other declining taxa present in yards treated with mosquito sprays.
Our calculations utilized acute contact LD50 values, which we used to investigate the acute risk to honey bees and butterfly caterpillars coated by insecticides when the sprays occurred. However, home mosquito treatments are often termed “barrier sprays”, and use larger droplet sizes that are designed to stick to vegetation around the perimeter of the yard and last for up to several weeks. These may expose insects to chronic contact toxicity concerns, from walking across or repeatedly landing on contaminated plants, and as a rule chronic toxicity levels are lower than acute toxicity levels. Additionally, these long-lived applications may cause acute and chronic oral toxicity to pollinators collecting pollen and nectar, and herbivores that feed on vegetation. Herbivores that are relatively stationary and unable to easily leave contaminated plants likely have increased risk. Because we were unable to examine chronic risk or dietary exposure, we are almost certainly underestimating the risks mosquito sprays pose to pollinators.
We used honey bees as a representative bee species for our exposure calculations because there is more toxicological data in the literature for honey bees when compared to native bees, as the species is used in regulatory testing. However, honey bee responses to pesticides are frequently not representative of other bee species (Arena and Sgolastra, 2014, Stoner, 2016, Shahmohamadloo et al., 2024), and mosquito sprays may be more toxic to various other bee species. Furthermore, solitary bees are unable to buffer the effects of pesticide exposure throughout a colony, and pesticide exposure has direct impacts on egg laying females when they are foraging.
There are several other ways that we have likely underestimated risk to pollinators from these sprays, depending on the method of exposure. For example, we have focused on the possibility of contact exposure, but caterpillars feeding on leaves of sprayed plants may encounter levels that trigger acute oral toxicity concerns. Given the fact that these treatments are designed to adhere to plant leaves and last ~21 days, the chronic oral toxicity may be significant. Additionally, we were unable to account for potential synergism between pesticide combinations, and our simple additive RQ of pyrethroids likely underestimates risk. Piperonyl butoxide, a synergist added to increase the toxicity of mosquito sprays, was consistently present across samples and can increase the toxicity of pyrethroids many times over, depending on the organism and pyrethroid (Joyce et al., 1988, Hagler et al., 1989, Reid et al., 2020). Even a more modest synergism would mean we are dramatically underestimating potential risk, since piperonyl butoxide was found on 67% of mosquito control district samples.
Furthermore, the other pesticides detected, beyond insecticides, can work additively or synergistically, increasing risk in yards with a higher pesticide richness. For example, fungicides have been found to increase pyrethroid toxicity in bumble bees (Iverson et al., 2019) and honey bees (Thompson and Wilkins, 2003), synergisms that we were unable to account for in our analyses. We also detected neonicotinoid and insect growth regulator (IGR) insecticides in hired yards. The IGRs may have been part of the barrier treatments (Stoops et al., 2019), and are designed to kill juvenile insects. The source of neonicotinoids is uncertain; with bands displayed for such short periods of time, the neonicotinoids may have been from residues in the application equipment.
This study evaluated single applications. However, these residential sprays are typically applied on a schedule. Some of our volunteers stated that these sprays start as early as February and go as late as November. Thus, the cumulative impact of these sprays on pollinators and other insects in the garden over the course of a year may be high, even in yards where we found detections at lower Risk Quotient values in our snapshot survey.
As this was an observational study, the landscape context differed between sites. Some participants lived across the street from the neighbor who sprayed, while others were on directly adjoining properties. Similarly, obstacles like fences and vegetation may have altered pesticide drift at some sites. Also, the practices of individual companies or applicators would influence deposition. In future studies, a larger sample size to account for some of these site-level variables would allow for better quantification of the relative impact of distance and common obstacles like shrubs and fencing on residues from pyrethroids and other pesticides. Yet another source of variation in our study comes from the likelihood that volunteers may not have always placed bands in the most optimal locations to collect mosquito sprays, in particular the four that hired spray services. These companies tend to spray vegetation in the yard, so the bands may not have been directly sprayed, leading to lower levels of pyrethroid deposition. On average, sprayed yards had ~117 ng/cm2 of pyrethroids per band. Previous studies have found higher amounts of bifenthrin on vegetation after barrier sprays, e.g. 305 – 1233 ng/cm2 (VanDusen et al., 2016). However, lower amounts have also been found, for example 22.4 – 72.0 ng/cm2 (Hoffmann et al., 2009). These variations may be due to sprayer type, pesticide formulation, and differences in the vegetation like surface area and composition of the waxy cuticle (McMillan et al., 2018).
Conclusion
Our intention was to increase understanding of the potential for mosquito sprays to contribute to the stressors insects face in residential areas. We recognize, of course, that mosquito-borne diseases are a genuine societal concern, and balancing the need to control those diseases while minimizing non-target impacts is an issue that deserves more attention. We hope that the results presented here encourage more attention being placed on the methods used in mosquito control, and the extent to which “adulticiding” is used as a primary control strategy. The most immediate actions can be taken by individuals choosing non-chemical strategies to avoid unwanted mosquito exposure: wearing protective clothing and dumping non-natural standing water once a week (CDC, 2024), which has no negative consequences for beneficial insects. Finally, our efforts to complete the risk analysis portion of this study highlight the lack of data about the impacts of insecticides to most non-Apis mellifera insect species. These data gaps lead to an inability to accurately assess the risk of pesticide applications to beneficial insects, and highlight the need for mandatory testing of terrestrial invertebrates beyond honey bees. While the breadth of risk is not clear, our findings clearly show that home sprays to control adult mosquitoes could kill or harm pollinators present in the area. These results also highlight the value of community-wide, targeted, and ecologically-sound management strategies, instead of relying on routine pesticide treatments.
Acknowledgments
We thank the volunteers who participated in this study. Thank you to A. Tran Lam and K. Enshayan with Good Neighbor Iowa and P. Helfrich with Bee City USA Decatur for help with data collection. We gratefully thank the Carroll Petrie Foundation for funding.
Author Contributions
Aaron Anderson: Conceptualization, project coordination, writing – original draft (lead), formal analysis (supporting), writing – review and editing
Aimee Code: Conceptualization, funding acquisition, writing – review and editing
Rosemary Malfi: Conceptualization, project coordination
Camille Holladay: Investigation, writing – original draft (supporting)
Matthew L. Forister: Conceptualization, formal analysis (lead), writing – review and editing
Data Availability
All data and code can be found here.
Supplementary Information
Supplementary information can be found here.
Transparent Peer Review
Results from the Transparent Peer Review can be found here.
Recommended Citation
Anderson, A., A. Code, R. Malfi, C. Holladay, and M.L. Forister. 2025. An assessment of risk to pollinators from mosquito control in residential settings. Stacks Journal: 25013. https://doi.org/10.60102/stacks-25013
References
Arena, M., and F. Sgolastra. 2014. A meta-analysis comparing the sensitivity of bees to pesticides. Ecotoxicology 23: 324–334.
Baldock, K. C., M. A. Goddard, D. M. Hicks, W. E. Kunin, N. Mitschunas, L. M. Osgathorpe, S. G. Potts, K. M. Robertson, A. V. Scott, G. N. Stone, and I. P. Vaughan. 2015. Where is the UK’s pollinator biodiversity? The importance of urban areas for flower-visiting insects. Proceedings of the Royal Society B: Biological Sciences 282: 20142849.
Bartomeus, I., J. S. Ascher, J. Gibbs, B. N. Danforth, D. L. Wagner, S. M. Hedtke, and R. Winfree. 2013. Historical changes in northeastern US bee pollinators related to shared ecological traits. Proceedings of the National Academy of Sciences 110: 4656–4660.
Bartoń, K. 2025. MuMIn: Multi-Model Inference. R package version 1.48.11. https://CRAN.R-project.org/package=MuMIn.
Brett, M. T. 2014. Resource Polygon Geometry Predicts Bayesian Stable Isotope Mixing Model Bias. Marine Ecology Progress Series 514: 1–12. https://doi.org/10.3354/meps11017.
Bates, D., M. Maechler, B. Bolker, S. Walker, R.H.B. Christensen, H. Singmann, B. Dai, G. Grothendieck, P. Green. and M.B. Bolker. 2015. Package ‘lme4’. Convergence 12: 2.
Bonds, J. A. S. 2012. Ultra-low-volume space sprays in mosquito control: a critical review. Medical and Veterinary Entomology 26: 121–130.
Cameron, S. A., J. D. Lozier, J. P. Strange, J. B. Koch, N. Cordes, L. F. Solter, and T. L. Griswold. 2011. Patterns of widespread decline in North American bumble bees. Proceedings of the National Academy of Sciences 108: 662–667.
Centers for Disease Control. 2024. Preventing mosquito bites. https://www.cdc.gov/mosquitoes/prevention/index.html. Accessed October 2024.
Cilek, J. E. 2008. Application of insecticides to vegetation as barriers against host-seeking mosquitoes. Journal of the American Mosquito Control Association 24: 172–176.
Colla, S. R., and L. Packer. 2008. Evidence for decline in eastern North American bumblebees (Hymenoptera: Apidae), with special focus on Bombus affinis Cresson. Biodiversity and Conservation 17: 1379–1391
Crone, E. E., and C. B. Schultz. 2021. Resilience or catastrophe? A possible state change for monarch butterflies in western North America. Ecology Letters 24: 1533–1538.
Decourtye, A., J. Devillers, S. Cluzeau, M. Charreton, and M. H. Pham-Delègue. 2004. Effects of imidacloprid and deltamethrin on associative learning in honeybees under semi-field and laboratory conditions. Ecotoxicology and Environmental Safety 57: 410–419.
Dittemore, C. M., A. Anderson, A. Code, A. Lenard, M. R. Douglas, C. A. Halsch, and M. L. Forister. 2025. Pesticides detected in two urban areas have implications for local butterfly conservation. Environmental Toxicology and Chemistry: vgaf218.
Domonoske, C. 2016. S.C. county sprays for mosquitoes but accidentally takes out millions of bees. https://www.npr.org/sections/thetwo-way/2016/09/02/492404411/s-c-county-sprays-for-mosquitos-but-accidentally-takes-out-millions-of-bees.
Hilderbrand, G. V., S. D. Farley, C. T. Robbins, T.A. Hanley, K. Titus, and C. Servheen. 1996. Use of Stable Isotopes to Determine Diets of Living and Extinct Bears. Canadian Journal of Zoology 74: 2080–2088. https://doi.org/10.1139/z96-236.
Edwards, C. B., E. F. Zipkin, E. H. Henry, N. M. Haddad, M. L. Forister, K. J. Burls, S. P. Campbell, E. E. Crone, J. Diffendorfer, M. R. Douglas, and R. G. Drum. 2025. Rapid butterfly declines across the United States during the 21st century. Science 387: 1090–1094.
Federal Register. 2024. Endangered and Threatened Species: Species Status with Section 4(d) Rule for Monarch Butterfly and Designation of Critical Habitat. Fed. Reg. 89(239): 100662–100716.
Fetridge, E. D., J. S. Ascher, and G. A. Langellotto. 2008. The bee fauna of residential gardens in a suburb of New York City (Hymenoptera: Apoidea). Annals of the Entomological Society of America 101: 1067–1077.
Fischer, J., T. Müller, A. K. Spatz, U. Greggers, B. Grünewald, and R. Menzel. 2014. Neonicotinoids interfere with specific components of navigation in honeybees. PLoS One 9: e91364.
Forister, M. L., C. A. Halsch, C. C. Nice, J. A. Fordyce, T. E. Dilts, J. C. Oliver, K. L. Prudic, A. M. Shapiro, J. K. Wilson, and J. Glassberg. 2021. Fewer butterflies seen by community scientists across the warming and drying landscapes of the American West. Science 371: 1042–1045.
Fulcher, A., M. Farooq, M. L. Smith, C. X. Li, J. M. Scott, E. Thomson, P. E. Kaufman, and R. D. Xue. 2015. Evaluation of a new spraying machine for barrier treatment and penetration of bifenthrin on vegetation against mosquitoes. Journal of the American Mosquito Control Association 31: 85–92.
Gill, R. J., and N. E. Raine. 2014. Chronic impairment of bumblebee natural foraging behaviour induced by sublethal pesticide exposure. Functional Ecology 28: 1459–1471.
Guzman, L. M., E. Elle, L. A. Morandin, N. S. Cobb, P. R. Chesshire, L. M. McCabe, A. Hughes, M. Orr, and L. K. M’Gonigle. 2024. Impact of pesticide use on wild bee distributions across the United States. Nature Sustainability 7: 1324–1334.
Halsch, C. A., A. Code, S. M. Hoyle, J. A. Fordyce, N. Baert, and M. L. Forister. 2020. Pesticide contamination of milkweeds across the agricultural, urban, and open spaces of low-elevation northern California. Frontiers in Ecology and Evolution 8: 162.
Hartig, F. 2024. DHARMa: Residual Diagnostics for Hierarchical (Multi-Level/Mixed) Regression Models. R package version 0.4.7. https://github.com/florianhartig/dharma.
Hoang, T. C., R. L. Pryor, G. M. Rand, and R. A. Frakes. 2011. Use of butterflies as nontarget insect test species and the acute toxicity and hazard of mosquito control insecticides. Environmental Toxicology and Chemistry 30: 997–1005.
Hoang, T. C., and G. M. Rand. 2015. Mosquito control insecticides: A probabilistic ecological risk assessment on drift exposures of naled, dichlorvos (naled metabolite), and permethrin to adult butterflies. Science of the Total Environment 502: 252–265.
Hoffmann, W. C., M. Farooq, T. W. Walker, B. Fritz, D. Szumlas, B. Quinn, U. Bernier, J. Hogsette, Y. Lan, Y. Huang, and V. L. Smith. 2009. Canopy penetration and deposition of barrier sprays from electrostatic and conventional sprayers. Journal of the American Mosquito Control Association 25: 323–331.
IUCN. 2024. The IUCN Red List of Threatened Species. Version 2024–2. https://www.iucnredlist.org. Accessed January 23, 2025.
Iverson, A., C. Hale, L. Richardson, O. Miller, and S. McArt. 2019. Synergistic effects of three sterol biosynthesis inhibiting fungicides on the toxicity of a pyrethroid and neonicotinoid insecticide to bumble bees. Apidologie 50: 733–744.
Jacobson, M. M., E. M. Tucker, M. E. Mathiasson, and S. M. Rehan. 2018. Decline of bumble bees in northeastern North America, with special focus on Bombus terricola. Biological Conservation 217: 437–445.
James, D. G., D. Nunnallee, and R. M. Pyle. 2011. Life histories of Cascadia butterflies. 447 pp. Corvallis: Oregon State University Press.
Jeran, N., M. Grdiša, F. Varga, Z. Šatović, Z. Liber, D. Dabić, and M. Biošić. 2020. Pyrethrin from Dalmatian pyrethrum (Tanacetum cinerariifolium): biosynthesis, biological activity, methods of extraction and determination. Phytochemistry Reviews: 1–31.
Katsuda, Y. 2011. Progress and future of pyrethroids. In Pyrethroids, edited by N. Matsuo and T. Mori. Topics in Current Chemistry 314. Springer, Berlin, Heidelberg. https://doi.org/10.1007/128_2011_252.
Kolde, R., and M. R. Kolde. 2015. Package “pheatmap.” R package version 1.7.790.
Krishnan, N., Y. Zhang, K. G. Bidne, R. L. Hellmich, J. R. Coats, and S. P. Bradbury. 2020. Assessing field-scale risks of foliar insecticide applications to monarch butterfly (Danaus plexippus) larvae. Environmental Toxicology and Chemistry 39: 923–941.
Krueger, A. J., K. Hanford, T. J. Weissling, A. M. Vélez, and T. D. Anderson. 2021. Pyrethroid exposure reduces growth and development of monarch butterfly caterpillars. Journal of Insect Science 21: 2.
Lewis, K. A., J. Tzilivakis, D. Warner, and A. Green. 2016. An international database for pesticide risk assessments and management. Human and Ecological Risk Assessment 22: 1050–1064.
Leuenberger, W., J. W. Doser, M. W. Belitz, L. Ries, N. M. Haddad, W. E. Thogmartin, and E. F. Zipkin. 2025. Three decades of declines restructure butterfly communities in the Midwestern United States. Proceedings of the National Academy of Sciences 122: e2501340122.
Lowenstein, D. M., K. C. Matteson, I. Xiao, A. M. Silva, and E. S. Minor. 2014. Humans, bees, and pollination services in the city: The case of Chicago, IL (USA). Biodiversity and Conservation 23: 2857–2874.
Narahashi, T., J. M. Frey, K. S. Ginsburg, and M. L. Roy. 1992. Sodium and GABA-activated channels as the targets of pyrethroids and cyclodienes. Toxicology Letters 64: 429–436.
Nelson, T. J. 2020. Monarch butterfly kill-off called horrible as questions arise about mosquito pesticide. https://www.kvrr.com/2020/09/08/monarch-butterfly-kill-off-called-horrible-as-questions-arise-about-mosquito-pesticide/.
Oberhauser, K. S., S. J. Brinda, S. Weaver, R. D. Moon, S. A. Manweiler, and N. Read. 2006. Growth and survival of monarch butterflies (Lepidoptera: Danaidae) after exposure to permethrin barrier treatments. Environmental Entomology 35: 1626–1634.
Oberhauser, K. S., S. A. Manweiler, R. Lelich, M. Blank, R. V. Batalden, and A. De Anda. 2009. Impacts of ultra-low volume resmethrin applications on non-target insects. Journal of the American Mosquito Control Association 25: 83–93.
O’Connell, S. G., L. D. Kincl, and K. A. Anderson. 2014. Silicone wristbands as personal passive samplers. Environmental Science & Technology 48: 3327–3335.
Pokhrel, V., N. A. DeLisi, R. G. Danka, T. W. Walker, J. A. Ottea, and K. B. Healy. 2018. Effects of truck-mounted, ultra low volume mosquito adulticides on honey bees (Apis mellifera) in a suburban field setting. PLoS One 13: e0193535.
Poquet, Y., L. Bodin, M. Tchamitchian, M. Fusellier, B. Giroud, F. Lafay, A. Buleté, S. Tchamitchian, M. Cousin, M. Pelissier, and J. L. Brunet. 2014. A pragmatic approach to assess the exposure of the honey bee (Apis mellifera) when subjected to pesticide spray. PLoS One 9: e113728.
Qualls, W., B. Moser, R. Pereira, and P. Koehler. 2022. Impacts of barrier insecticide mixtures on mosquito, Aedes aegypti and non-target honey bee, Apis mellifera. Journal of the Florida Mosquito Control Association 69: 34–42.
Raine, N. E., and M. Rundlöf. 2024. Pesticide exposure and effects on non-Apis bees. Annual Review of Entomology 69: 551–576.
Raven, P. H., and D. L. Wagner. 2021. Agricultural intensification and climate change are rapidly decreasing insect biodiversity. Proceedings of the National Academy of Sciences 118: e2002548117.
Richardson, L. L., K. P. McFarland, S. Zahendra, and S. Hardy. 2019. Bumble bee (Bombus) distribution and diversity in Vermont, USA: a century of change. Journal of Insect Conservation 23: 45–62.
Sandrock, C., L. G. Tanadini, J. S. Pettis, J. C. Biesmeijer, S. G. Potts, and P. Neumann. 2013. Sublethal neonicotinoid insecticide exposure reduces solitary bee reproductive success. Agricultural and Forest Entomology 16: 119–128.
Shahmohamadloo, R. S., M. L. Tissier, and L. M. Guzman. 2024. Risk assessments underestimate threat of pesticides to wild bees. Conservation Letters 17: e13022.
Siviter, H., G. L. Pardee, N. Baert, S. McArt, S. Jha, and F. Muth. 2023. Wild bees are exposed to low levels of pesticides in urban grasslands and community gardens. Science of the Total Environment 858: 159839.
Stoner, K. A. 2016. Current pesticide risk assessment protocols do not adequately address differences between honey bees (Apis mellifera) and bumble bees (Bombus spp.). Frontiers in Environmental Science 4: 79.
Stoner, K. A., R. S. Cowles, A. Nurse, and B. D. Eitzer. 2019. Tracking pesticide residues to a plant genus using palynology in pollen trapped from honey bees (Hymenoptera: Apidae) at ornamental plant nurseries. Environmental Entomology 48: 351–362.
Stoops, C. A., W. A. Qualls, T. V. T. Nguyen, and S. L. Richards. 2019. A review of studies evaluating insecticide barrier treatments for mosquito control from 1944 to 2018. Environmental Health Insights 13: 1178630219859004.
Tai, Z., C. R. Connelly, S. Kuczynski Lange, N. Foley, J. De Leon Rivera, S. Lozano, and R. J. Nett. 2024. A scoping review to determine if adverse human health effects are associated with use of organophosphates for mosquito control. Journal of Medical Entomology 62: 8–18.
Theodorou, P., R. Radzevičiūtė, G. Lentendu, B. Kahnt, M. Husemann, C. Bleidorn, J. Settele, O. Schweiger, I. Grosse, T. Wubet, and T. E. Murray. 2020. Urban areas as hotspots for bees and pollination but not a panacea for all insects. Nature Communications 11: 576.
Thompson, H., and S. Wilkins. 2003. Assessment of the synergy and repellency of pyrethroid/fungicide mixtures. Bulletin of Insectology 56: 131–134.
United States Environmental Protection Agency. 2014. Guidance for assessing pesticide risks to bees. https://www.epa.gov/sites/default/files/2014-06/documents/pollinator_risk_assessment_guidance_06_19_14.pdf. Accessed October 2024.
United States Environmental Protection Agency. 2011. Appendix D. Risk Quotient (RQ) and Levels of Concern (LOC). https://www3.epa.gov/pesticides/endanger/litstatus/effects/redlegfrog/endosulfan/appendix-d.pdf Accessed October 2024.
VanDusen, A. E., S. L. Richards, and J. A. G. Balanay. 2016. Evaluation of bifenthrin barrier spray on foliage in a suburban eastern North Carolina neighborhood. Pest Management Science 72: 1004–1012.
Wagner, D. L., E. M. Grames, M. L. Forister, M. R. Berenbaum, and D. Stopak. 2021. Insect decline in the Anthropocene: Death by a thousand cuts. Proceedings of the National Academy of Sciences 118: e2023989118.
Ward, L. T., M. L. Hladik, A. Guzman, S. Winsemius, A. Bautista, C. Kremen, and N. J. Mills. 2022. Pesticide exposure of wild bees and honey bees foraging from field border flowers in intensively managed agriculture areas. Science of the Total Environment 831: 154697.
Wenzel, A., I. Grass, V. V. Belavadi, and T. Tscharntke. 2020. How urbanization is driving pollinator diversity and pollination–A systematic review. Biological Conservation 241: 108321.
Wepprich, T., J. R. Adrion, L. Ries, J. Wiedmann, and N. M. Haddad. 2019. Butterfly abundance declines over 20 years of systematic monitoring in Ohio, USA. PLoS One 14: e0216270.
Accepted by 3 of 3 reviewers
Open Access
Peer-Reviewed
Creative Commons
Submitted: 21 March 2025
Accepted: 10 July 2025
Published: 11 December 2025
Carroll Petrie Foundation
Conflicts of Interest:
The authors declares no conflicts of interest.








